Understanding defects paves the way for longer lifetimes for sodium-ion batteries -- and lower energy storage costs.
October 16, 2023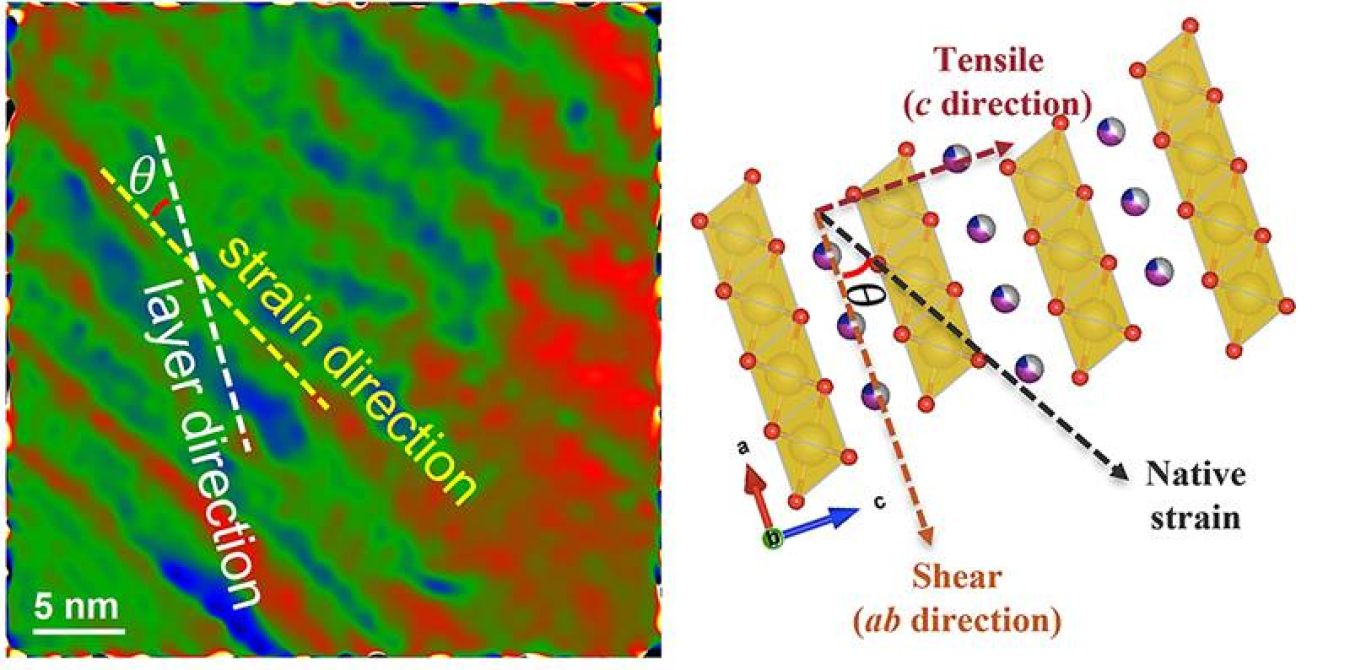
The Science
Lithium-ion batteries are the standard for electric vehicles, but their raw materials are costly and can have unreliable supply chains. Sodium-ion batteries are an alternative that could alleviate some of these challenges. However, the performance of these batteries declines rapidly with repeated charges and discharges. This is a major barrier to bringing these devices to the marketplace. In this study, researchers used a combination of electron microscopy and X-ray scattering to find a cause of this decline in performance: defects introduced in making the cathode material. This knowledge will help researchers design better cathodes for longer-lasting sodium-ion batteries.
The Impact
Using the insights gained from this study, battery developers may be able to create cathodes for sodium-ion batteries with virtually no defects. These new devices could cost less than current lithium-based batteries and have longer lifetimes. This new technology could lead to more affordable electric vehicles with longer driving ranges and faster charging times. Less expensive batteries could also lead to lower costs for energy storage on the electric grid.
Summary
The key to this research, by a team from Argonne National Laboratory, University of Wisconsin-Milwaukee, and Stanford University, was to combine different experimental techniques. The study examined the newly synthesized cathode materials using research tools at two Department of Energy (DOE) Office of Science user facilities: high-energy X-ray beams at the Advanced Photon Source and the analytical capabilities of the Center for Nanoscale Materials. The synthesis process involves slowly heating the cathode materials before rapidly dropping the temperature. Using transmission electron microscopy and surface X-ray diffraction to examine this material during this process in situ, scientists concluded that defects formed during the cooling-off period. These defects cause cracking of the cathode particles and a decline in performance, which only gets worse when cathodes are charged quickly or at high temperatures. Eventually, this can result in a “structural earthquake” in the cathode, leading to catastrophic battery failure.
Armed with this knowledge, battery developers can adjust the conditions during battery synthesis and control the defects in sodium-ion battery cathodes. This work leverages the capability of both user facilities to capture real-time information on transformations in materials as they happen, under controllable changes in the sample environment. These findings highlight the importance of eliminating these defects to ensure long-term stable cycling of sodium-ion batteries at higher voltages.
Contact
Guiliang Xu
Argonne National Laboratory
xug@anl.gov
Funding
Funding of this research was provided by the DOE Vehicle Technologies Office. Work was performed at the Advanced Photon Source and the Center for Nanoscale Materials, both DOE Office of Science user facilities.
Publications
Xu, GL., et al., Native lattice strain induced structural earthquake in sodium layered oxide cathodes. Nature Communications 13, 436 (2022). [DOI: 10.1038/s41467-022-28052-x]
Related Links
Pivotal Battery Discovery could Impact Transportation and the Grid, Argonne National Laboratory News

