The distribution of indium and thallium between the aqueous and organic phases is the key to understanding the separation of these elements. An
February 1, 2017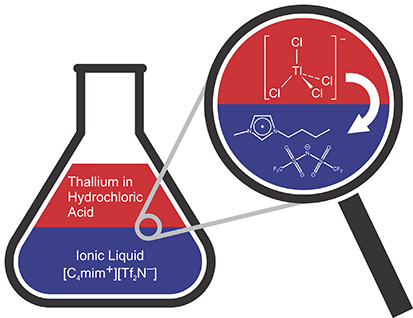
The Science
The chemical properties of "super heavy" element 113 are almost completely unknown, so a team of researchers from the Cyclotron Institute at Texas A&M University and the Institut Pluridisciplinaire Hubert Curien in France are developing techniques that could be used to study this fleeting element. As part of that effort, they are comparing the properties of element 113 to the chemically similar elements (indium and thallium); to do so, the team studied the separation of these two elements using a new class of "designer molecules" called ionic liquids.
The Impact
Measuring the chemical properties of element 113 and other "super heavy" elements will increase our understanding of the principles that control the Periodic Table. Comparing the data from element 113 to results for similar elements, obtained using the team's fast, efficient, single-step process, reveals trends that arise from the structure of the Periodic Table. This research could also lead to better methods of re-using indium, a metal that is part of flat-panel displays but is not currently mined in the United States.
Summary
The distribution of indium and thallium between the aqueous and organic phases is the key to understanding the separation of these elements. An aqueous solution, containing trace amounts of the metals dissolved in hydrochloric acid, is put in contact with an ionic liquid. The latter is a class of non-toxic "organic salts" that are liquid at room temperature, in contrast with traditional salts, which are solids. The acid and the ionic liquid do not mix, like oil and water, so measuring which phase contains a certain metal can reveal its behavior and chemical form under these conditions. Thallium is preferentially found in the ionic liquid, and a systematic study of the reaction mechanism has shown that the key step is to convert thallium from its 1+ oxidation state to its 3+ oxidation state. This new technique could potentially be applied to the study of element 113 (which was recently recognized by the International Union of Pure and Applied Chemistry, the body that decides on the validity of elements) to help determine how many oxidation states it has, which is currently unknown.
Contact
Charles M. Folden III
Texas A&M University
Folden@comp.tamu.edu
Evgeny E. Tereshatov
Texas A&M University
ETereshatov@tamu.edu
Funding
This material is based upon work supported by the U.S. Department of Energy, Office of Science, Office of Nuclear Physics under award DE-FG02-12ER41869/DE-SC0008126. Additionally, this work was supported by the Robert A. Welch Foundation under award A-1710 and funding provided by Lawrence Livermore National Laboratory to support collaborative research at Texas A&M University. Finally, this work was supported by the Centre National de la Recherche Scientifique of France (CNRS) and the University of Strasbourg.
Publications
E.E. Tereshatov, M. Yu. Boltoeva, V. Mazan, M.F. Volia, and C.M. Folden III, "Thallium transfer from hydrochloric acid media into pure ionic liquids ," Journal of Physical Chemistry B 120(9), 2311-2322 (2016). [DOI: 10.1021/acs.jpcb.5b08924].
," Journal of Physical Chemistry B 120(9), 2311-2322 (2016). [DOI: 10.1021/acs.jpcb.5b08924].
Related Links
Texas A&M University webpage: Cyclotron Institute
Highlight Categories
Program: NP
Performer/Facility: University
Additional: Collaborations, International Collaboration

