The transition state is where reactants become products in a chemical reaction. Defined as the highest energy point on the minimum energy path
February 1, 2017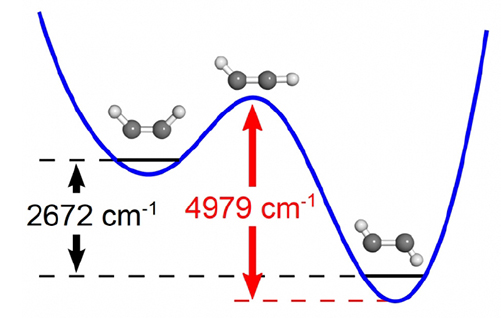
The Science
For the first time, chemists have devised a way to measure the energy of a chemical reaction's transition state—a fleeting, unstable tipping point. The transition state is like a mountain pass. Reactions require energy to cross it. The new technique also reveals information about the structure and vibrations of the transition state. Knowing the structure and vibration gives scientists insight into how a reaction really works.
The Impact
Transition states have always been regarded as impossible to study directly. Now, researchers can precisely determine the energy involved. Knowing that energy is important wherever chemistry is important, whether that's improving a car's gas mileage or a battery's longevity.
Summary
The transition state is where reactants become products in a chemical reaction. Defined as the highest energy point on the minimum energy path between products and reactants, it is central to chemical kinetics because the energy and geometric structure of this transition state are essential to both qualitative pictures and quantitative calculations of temperature-dependent reaction rates. However, due to the sub-picosecond transit time through the transition state region, direct quantitative experimental characterization of a transition state was thought to be impossible. Researchers at the Massachusetts Institute of Technology have developed a spectroscopic method by which the energy and structure of the transition state can be determined. This method is based on observing a signature pattern of vibrational energy levels in which the energy and structure of the transition state are encoded. Using two reactions as test cases, the cis-trans isomerization of electronically excited acetylene and the 1,2-hydrogen shift of HCN to HNC, the researchers collected data on transition state energies, structures, and vibrational frequencies, all of which closely matched theoretical predictions. This new method for studying transition states could alter our foundational understanding of reaction mechanisms and provide a new source of information about the rates of chemical reactions.
Contact
Robert W. Field
MIT
rwfield@mit.edu
Funding
This research was supported by a National Science Foundation Graduate Research Fellowship DGE 1144083 (P.B.C.), an Alexander von Humboldt Foundation Feodor Lynen fellowship for experienced researchers (G.Ch.M.), and the U.S. Department of Energy, Office of Science, Office of Basic Energy Sciences, Division of Chemical Sciences, Geosciences, and Biosciences through grant DE-FG0287ER13671.
Publications
J.H. Baraban, D.A. Matthews, and J.F. Stanton, "Communication: An accurate calculation of the S1 C2H2 cis-trans isomerization barrier height ." Journal of Chemical Physics 144, 111102 (2016). [DOI: 10.1063/1.4943865]
." Journal of Chemical Physics 144, 111102 (2016). [DOI: 10.1063/1.4943865]
J.H. Baraban, P.B. Changala, G.Ch. Mellau, J.F. Stanton, A.J. Merer, and R.W. Field, "Spectroscopic characterization of isomerization transition states ." Science 350, 1338 (2015). [DOI: 10.1126/science.aac9668]
." Science 350, 1338 (2015). [DOI: 10.1126/science.aac9668]
Related Links
Scientific American press release: For the First Time Chemists Measure the Energy of a Chemical Reaction's Transition State
MIT press release: MIT Chemists Characterize a Chemical State Thought To Be Unobservable
Science News article: Elusive Chemical Reaction Transition State Captured
Phys.org article: Chemical State Thought To Be Unobservable Characterized
EurekAlert! post: MIT Chemists Characterize a Chemical State Thought To Be Unobservable
Science Daily article: Chemists Characterize a Chemical State Thought To Be Unobservable
eScience News article: MIT Chemists Characterize a Chemical State Thought To Be Unobservable
Chemistry World article: Getting the Measure of Transition States
Highlight Categories
Additional: Collaborations, Non-DOE Interagency Collaboration

