When metal oxide nanoparticles are manufactured by reaction of gases at high temperature, they can form aggregates with complex geometric shapes.
June 9, 2016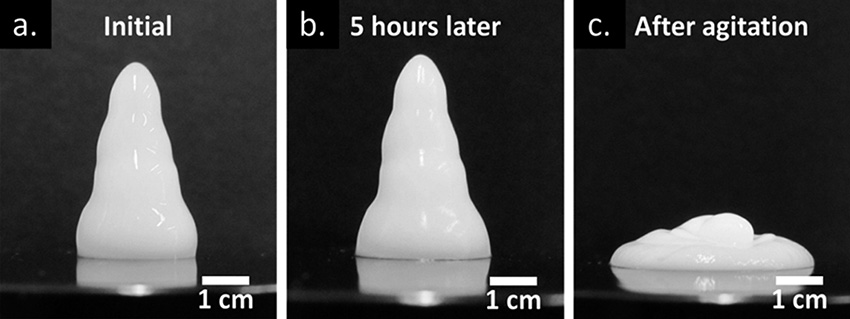
The Science
Gels that help prevent oppositely charged nanoparticles from settling out of solution enable applications from ceramic synthesis to adsorption of water and other compounds. Scientists carefully studied these reactions and the conditions, including controlled agitation, under which the gel collapses. They mapped out a detailed mechanistic understanding of the gel, revealing contributions from three district phenomena.
The Impact
The understanding gained from this research strengthens the ability to tune the properties of nanoparticle gels, which could enable broader applications in 3D printing, paints, gel-based consumer products, and geological surveying of oil reservoirs.
Summary
When metal oxide nanoparticles are manufactured by reaction of gases at high temperature, they can form aggregates with complex geometric shapes. Dispersions of these nanoparticles are commonly used as pigments and additives that adjust the viscosity of solutions for various technological applications ranging from timed-released chemical delivery to specialty surface coatings. Now, researchers in Oklahoma have made "pseudosolid" gels composed of these non-spherical particles. The structural integrity of the gel is due to electrostatic interactions between the mixed nanoparticles within the solution, some of which are negatively charged, others are positively charged. The gel formation depends on the ratio of particle sizes, their concentrations, and the acidity or basicity of the water in which the nanoparticles are mixed. In this research, their mechanical properties and structures were characterized by advanced spectroscopic and microscopic analyses. The resulting gels exhibited extreme "shear thinning" behavior; that is, the gels become less viscous when shear pressure is applied by vibrating the gels. This process measures the force required to separate the oppositely charged nanoparticles. Three phenomena governed their flow behavior: (1) the balance of electrostatic attractions and repulsions that prevent sedimentation and produce a gel network, (2) the molecular structure of the gel inherently creating a rigid network that results in a yield stress barrier, and (3) the hydration forces that resist the desorption of water and prevent dispersed particles from contacting one another. With an advanced fundamental understanding of these gels, improvements to gel-based products ranging from 3D printing to contrast agents in geological surveys of oil reservoirs may be on the horizon.
Contact
Daniel Resasco
D. and H. Bourne Chair and George Lynn Cross Professor Chemical, Biological, and Materials Engineering, University of Oklahoma
resasco@ou.edu
Funding
This work was supported by the U.S. Department of Energy, Office of Science, Office of Basic Energy Sciences.
Publications
J. Weston, D. Venkataramani, C. Aichele, B. Grady, J. Harwell, and D. Resasco, "Pseudosolid, shear-thinning gel formation in binary dispersions of metal oxide nanoparticles at low volume fractions ." Langmuir 49, 14982 (2014). [DOI: 10.1021/la503442a].
." Langmuir 49, 14982 (2014). [DOI: 10.1021/la503442a].
Highlight Categories
Performer/Facility: University

