Bulk metallic glasses are a class of metals that lack an ordered crystalline structure. This feature leads to unique properties, but it also
December 12, 2017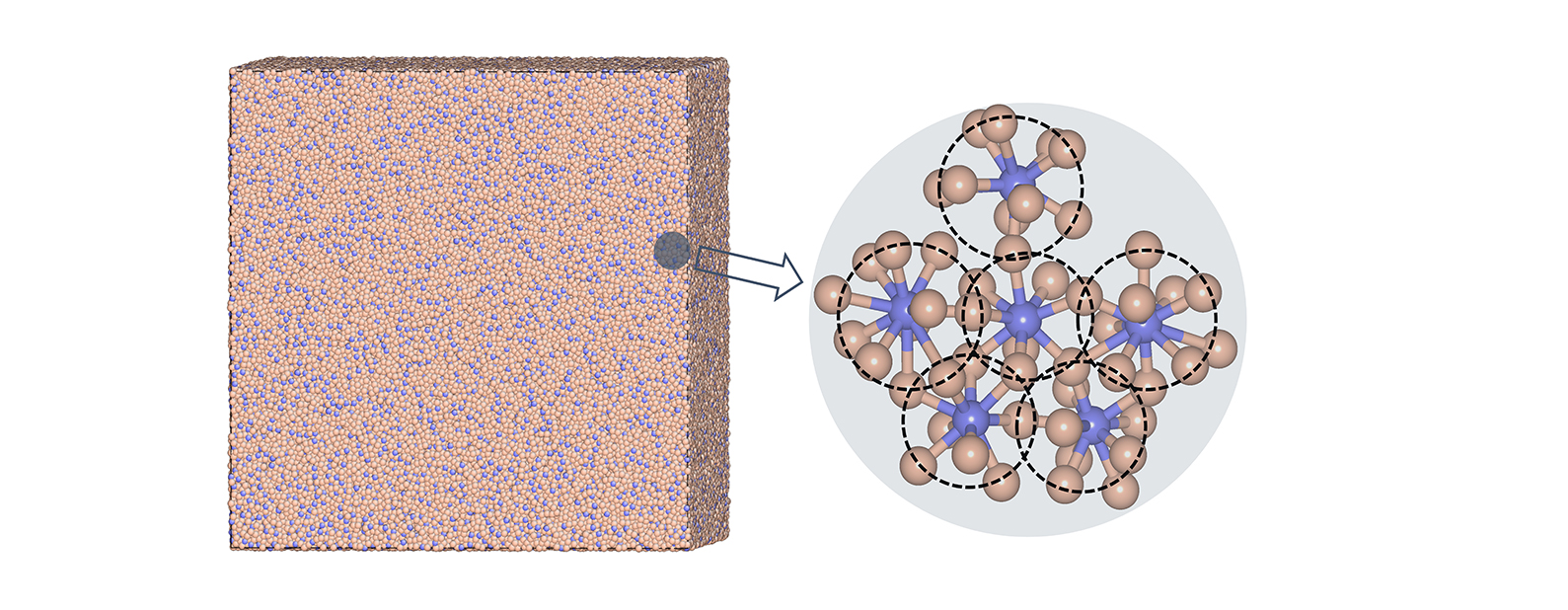
The Science
Stronger than steel yet easily fabricated, bulk metallic glasses are metals that lack an ordered atomic crystalline structure. The mystery of how the atoms are packed in these glasses has been studied for decades. Now, recent computer experiments have resolved a debate about atomic packing over a range of length scales. The simulations revealed the structure at various length scales is not similar.
The Impact
Elucidating atomic packing clarifies a long-standing debate over the structure of bulk metallic glasses. The results enhance our understanding of the properties of these glasses and other randomly ordered solids. For example, the work better explains key features such as how the metallic glass changes properties with temperature, and how it reacts to stress. Also, this work benefits scientists working to design high-strength metallic glasses.
Summary
Bulk metallic glasses are a class of metals that lack an ordered crystalline structure. This feature leads to unique properties, but it also results in large variability of properties that is not predictable. Also, the lack of order generally leads to poor resistance to sharp impacts (low toughness). It is well established that these materials possess local order on atomic length scales, but how this order manifests at larger "medium range" scales is an area of continuing investigation. One theory is that the atomic packing is "fractal"; that is, the structures/clusters of atoms are similar at a variety of length scales (much like a small rivulet in the mud has the same structure as the huge Mississippi River delta). Understanding precisely how that order can be described is important for predicting properties and establishing new directions to enable designing the physical and mechanical behavior of the metallic glasses. In this study, the researchers performed large-scale atomic computer simulations of a variety of metallic glass alloy compositions to make precise measurements of both the separation of individual atoms as well as the separation of clusters. Performing this analysis with such a large set of simulation data allowed measurements over a larger range of length scales. It revealed that the packing was not fractal. The new results also pinpointed challenges inherent in drawing conclusions about the atomic structure of these multicomponent systems using scattering-based experiments. The results illustrated features associated with the uniform deformation that can guide future analyses. Further, the researchers categorized at what level the small structural units were connected, which will help set the stage for further analysis of the unusual behavior of bulk metallic glasses.
Contact
Rob Ritchie
Lawrence Berkeley National Laboratory
roritchie@lbl.gov
Funding
This work was supported by the U.S. Department of Energy (DOE), Office of Science, Basic Energy Sciences and the National Energy Research Scientific Computing Center, a DOE Office of Science user facility.
Publications
J. Ding, M. Asta, and R.O. Ritchie, "On the question of fractal packing structure in metallic glasses ." Proceedings of the National Academy of Sciences USA 114, 8458-8463 (2017). [DOI: 10.1073/pnas.1705723114]
." Proceedings of the National Academy of Sciences USA 114, 8458-8463 (2017). [DOI: 10.1073/pnas.1705723114]
Related Links
Lawrence Berkeley National Laboratory research highlight: The Fractal Packing Structure in Metallic Glasses
Highlight Categories
Performer/Facility: DOE Laboratory, SC User Facilities, ASCR User Facilities, NERSC

