Ligands allow fine tuning of nanoparticle superstructure properties.
December 3, 2019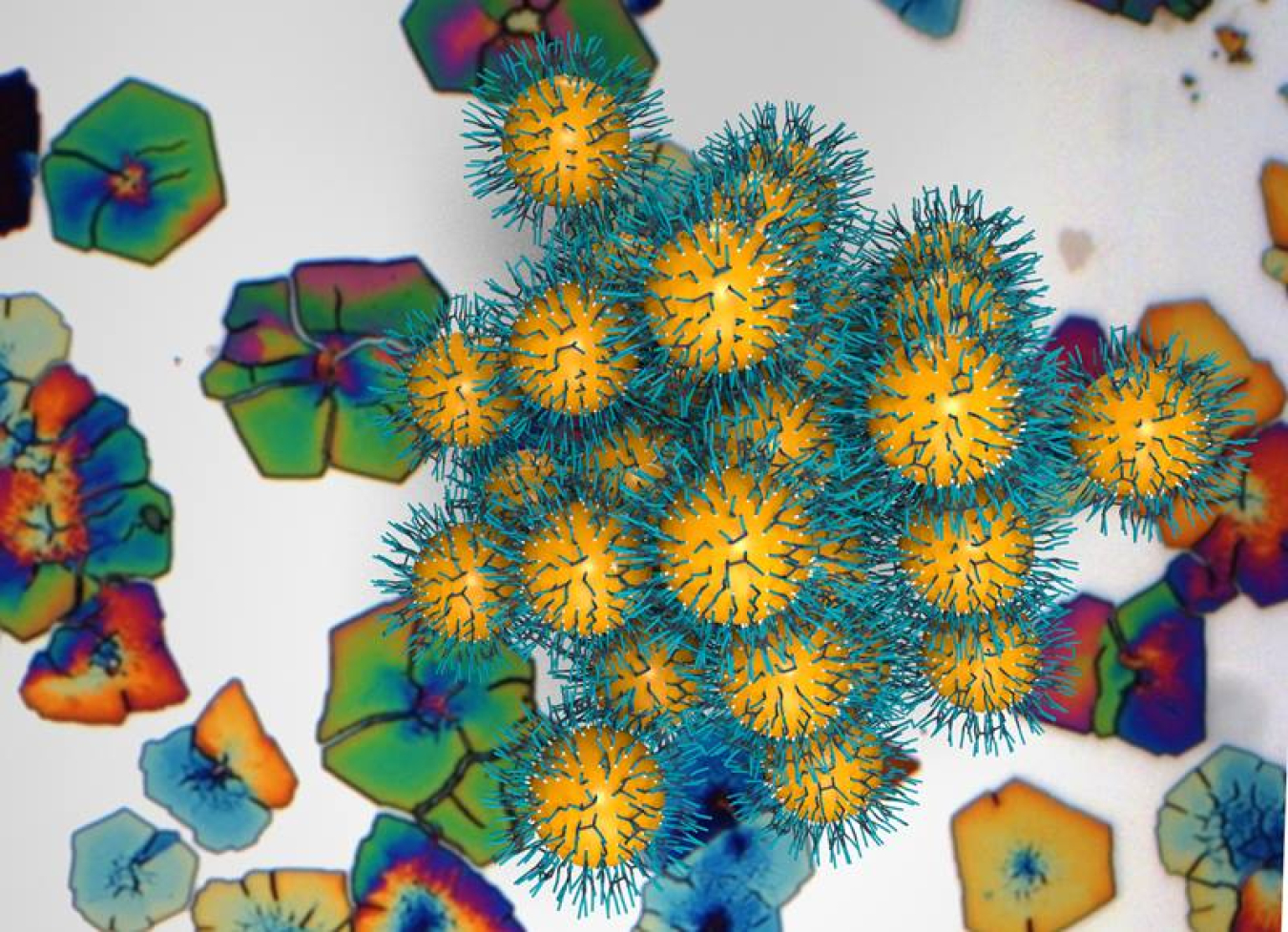
The Science
Nanoparticles are measured in nanometers, which are one billionth of a meter long. Nanoparticle superlattices are a type of group of these particles. This type of group has properties that are different from both the individual particles and the bulk material. Ions and molecules called ligands bind particles together in these superlattices. This new research shows how the ligands affect key structural and mechanical properties of the superlattices. The relationship between the number of binding ligands, how much they move on a nanoparticle surface, and how they move in the spaces between nanoparticles determine these properties.
The Impact
Nanoparticle superlattices have many useful properties. These include being able to switch from a metal to an insulator, having molecules that move together at specific frequencies, and forming permanent magnets. These materials may be useful in a number of applications, including sensors and solar cells. This discovery will help scientists better control properties of these superlattices. This knowledge is crucial for scientists to create next-generation materials for energy technologies.
Summary
To explore dynamic processes at the molecular level, faceted NPSL analogues of atomic crystals were synthesized in a toluene solution and characterized using scanning electron microscopy (SEM) at CNM, characterized using in situ small angle X-ray scattering techniques at APS, and modeled using molecular dynamics simulations in part at CNM. The nanoparticle superlattices consist of lead sulfide nanoparticles with capping oleic acid ligands. The results show that ligand coverage density dictates several key factors: the extent of diffusion of ligands over the nanoparticle surfaces, the spatial distribution of the ligands in the interstitial spaces between neighboring nanoparticles, and the fraction of ligands that interdigitate across different nanoparticles. Below a critical coverage density, the nanoparticle superlattices collapse to form disordered aggregates, even under ambient conditions. Above that coverage density, the nanoparticle superlattices surprisingly preserve their crystalline order even under very high applied pressures (∼40–55 GPa). Also, the interparticle spacing decreases with the applied pressure but returns to the original spacing upon pressure release. The combined results suggest the possibility of reversibly manipulating the lattice spacing of nanoparticle superlattices, and in turn, finely tuning their collective electronic, optical, thermo-mechanical, and magnetic properties.
Contact
Elena V. Shevchenko and Subramanian K.R.S. Sankaranarayanan Center for Nanoscale Materials, Argonne National Laboratory eshevchenko@anl.gov and ssankaranarayanan@anl.gov
Badri Narayanan, previously of Argonne, now at University of Louisville, badri.narayanan@louisville.edu
Funding
This research received support from Argonne National Laboratory through a Laboratory Directed Research and Development grant. Use of the Center for Nanoscale Materials and Advanced Photon Source, both Office of Science user facilities, was supported by the U.S. Department of Energy (DOE), Office of Science, Office of Basic Energy Sciences. This research used resources of the National Energy Research Scientific Computing Center and the Argonne Leadership Computing Facility, which are supported by the DOE Office of Science. An award of computer time was provided by the Innovative and Novel Computational Impact on Theory and Experiment (INCITE) program. Additional support came from the GeoSoilEnviroCARS program supported by the National Science Foundation—Earth Sciences and DOE.
Publications
T. Patra, H. Chan, P. Podsiadlo, E. Shevchenko, S. Sankaranarayanan, and B. Narayanan, “Ligand dynamics control structure, elasticity, and high-pressure behavior of nanoparticle superlattices,” Nanoscale 11, 10655 (2019); [DOI: 10.1039/c8nr09699f]

