Findings could rewrite textbooks about molecular structure for solvent ubiquitous in chemistry and biology.
January 22, 2019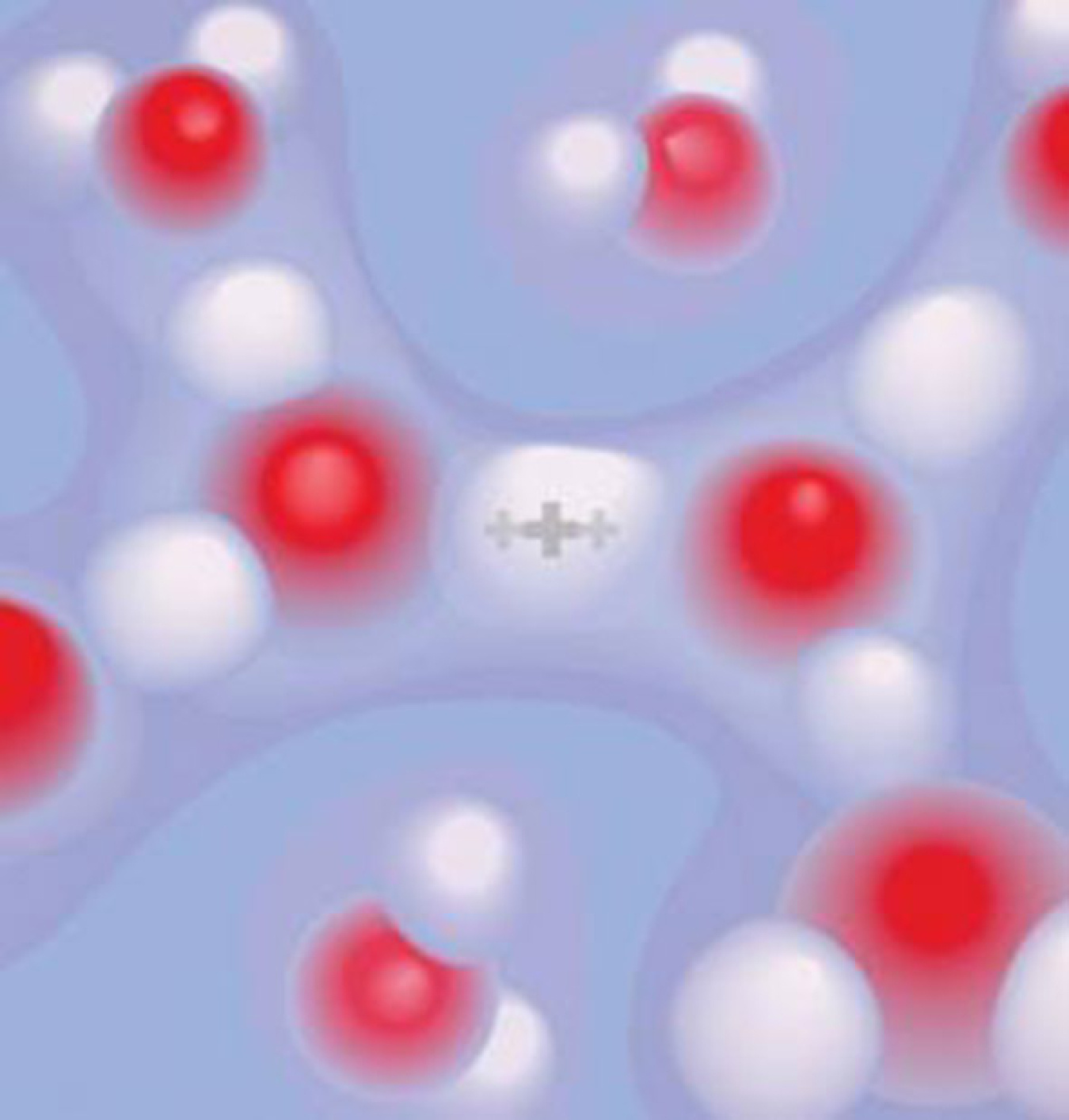
The Science
General chemistry textbooks teach that when acids (such as HCl) dissociate in water, the resulting proton associates with water as a hydronium ion (H3O+). It's a fundamental reaction. Using the latest ultrafast spectroscopy tools, researchers found that the proton is actually bound strongly between two water molecules. They also found that many structural variations are possible, and the structures persist longer than previously thought.
The Impact
Water is a ubiquitous solvent. It's present in biological organisms and industrial processes. Discoveries about the molecular structure of water during aqueous chemical reactions could have a broad impact. Examples range from our microscopic understanding of cellular respiration to energy storage and production. The problem is that the structures in solution are very complex, and the speed of structural changes is often too fast to measure. Researchers used a new ultrafast experimental approach to verify the path of protons in water.
Summary
Water is the universal solvent, important in biology, geology, and industrial processes. For some time, computational simulations suggested that aqueous protons didn't behave as described in general chemistry textbooks. But the protons were tough to study. They associate strongly with water molecules and structurally diffuse at rates that were too fast to follow in real time. Researchers took ultrafast snapshots of protons using 2-D vibrational spectroscopy. The technique was based on a new source of infrared light with bursts of energy only 50 femtoseconds in duration. The bursts occurred across an unusually wide region of the infrared spectrum, allowing many molecular structures to be observed instantly in water. Analyses of the data showed protons strongly bond to two waters in a fluctuating complex. There was no sign of the textbook hydronium ion (H3O+). Instead, measurements were consistent with a protonated water complex: the "Zundel-like" H5O2+. The data will benefit future computational models of transient water structure. Because of the critical role of the aqueous excess proton in redox chemistry, the data could also be important to a range of industrial reactions, including those in energy storage and production of fuels from water.
Contact
Andrei Tokmakoff
University of Chicago
tokmakoff@uchicago.edu
Funding
The Chemical Sciences, Geosciences, and Biosciences Division, Office of Basic Energy Sciences, Office of Science, Department of Energy funded this research. J.A.F. thanks the Arnold O. Beckman Foundation for support through a postdoctoral fellowship. N.H.C.L. acknowledges support from the Yen Fellowship programme.
Publications
J.A. Fournier, W.B. Carpenter, N.H.C. Lewis, and A. Tokmakoff, "Broadband 2D IR spectroscopy reveals dominant asymmetric H5O2+ proton hydration structures in acid solutions." Nature Chemistry 10, 932 (2018). [DOI: 10.1038/s41557-018-0091-y]
Related Links
University of Chicago press release: Proton Hydration Structures are Asymmetric, Study Reveals

