New finding may help explain why we develop chronic diseases and cancer as we age…and even why food decomposes over time.
May 7, 2020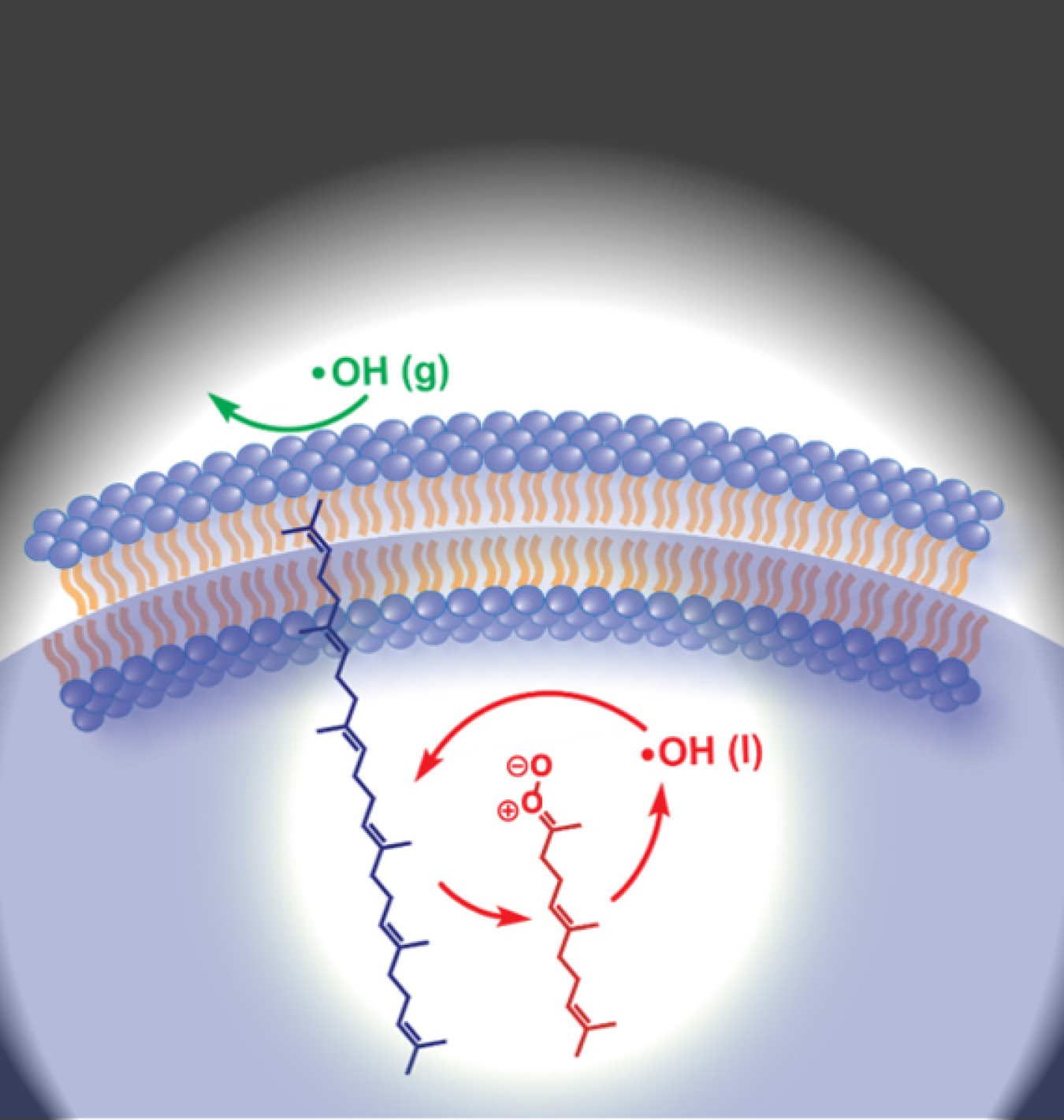
The Science
Scientists have identified a new mechanism that can cause the molecular building blocks of cell membranes to break down. The mechanism is based on a reaction cycle called autoxidation. This reaction results from the interaction of oxygen and free radicals of hydroxyl. Hydroxyl and other free radicals are atoms, molecules, and ions that are highly reactive. The new research shows that hydroxyl radicals, working in concert with Criegee intermediates (CI), can start a chain reaction. CIs are a type of molecule formed by reaction of certain hydrocarbons and ozone in the atmosphere.
The Impact
Autoxidation is a chain reaction responsible for the breakdown of organic molecules. It can affect biological cells, foods, plastics, petrochemicals, and fuels. The newly identified mechanism involving hydroxyl radicals and CIs means autoxidation may be much more widespread than previously understood. For example, it could explain why food becomes rancid. The finding is important in part because OH can form from smoke and industrial pollution.
Summary
Cell walls are built of organic molecules. One of the most important is a family of molecules called unsaturated lipids. Scientists have known for decades that these unsaturated lipid molecules break down over time due to autoxidation. Autoxidation is kicked off by oxygen and hydroxyl radicals (highly reactive, short-lived molecules). Over time, damage to unsaturated lipids in cells from autoxidation increases likelihood of cancer and other chronic age-related diseases. The prevailing autoxidation mechanism of unsaturated lipids such as those that make up cell membranes has been studied since the 1940s. It involves chain reactions initiated and propagated by hydrogen atoms and peroxy free radicals. Researchers have now identified a new autoxidation mechanism. This mechanism proceeds instead via hydroxyl radicals interacting with CIs. Researchers say it appears that as hydroxyl causes the autoxidation of unsaturated lipids, the process generates CIs, which in turn create more hydroxyl and so on to form a chain reaction. Although CIs are routinely encountered in atmospheric and synthetic organic chemistry, the researchers did not expect them to have a role in lipid autoxidation. The research results suggest that CIs play a much more prominent and general role in the chemistry of cells, food, petrochemicals, and the environment beyond what is currently believed.
The research included experiments on model lipid nanodroplets at the Advanced Light Source using vacuum ultraviolet aerosol mass spectrometry. The experiments revealed distinctive mass spectrometer signatures that indicate the presence and subsequent chain reaction chemistry involving the coupling of OH reactions with CIs.
Contact
Kevin R. Wilson
Senior Scientist and Deputy Director for Science Chemical Sciences Division Lawrence Berkeley National Laboratory
Email: krwilson@lbl.gov
Phone: 510-495-2474
Funding
This work was supported by the Gas Phase Chemical Physics program in the Chemical Sciences Geosciences and Biosciences Division of the Office of Basic Energy Sciences of the U.S. Department of Energy Office of Science. This research used resources of the Advanced Light Source, a DOE Scientific User Facility.
Publications
Meirong, Z., Heine, N., & Wilson, K.R., Evidence that Criegee intermediates drive autoxidation in unsaturated lipids, Proceedings of the National Academy of Sciences, Feb 2020. [DOI: 10.1073/pnas.1920765117]
Related Links
Berkeley Lab: Scientists Discover New Clue Behind Age-Related Diseases and Food Spoilage

