Magnesium metal anodes display improved cycling and temperature performance capabilities for rechargeable magnesium batteries.
October 29, 2019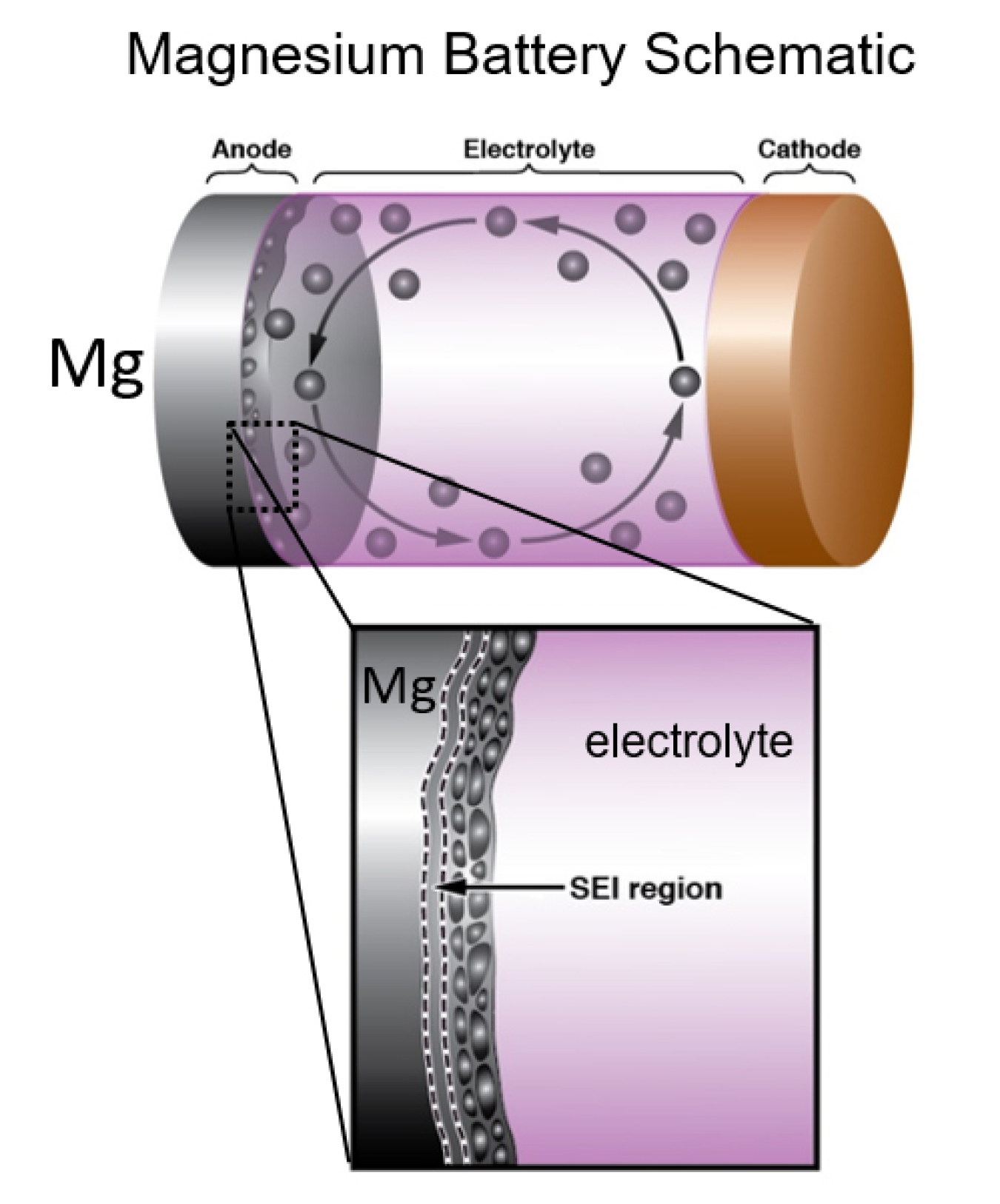
The Science
Researchers recently studied rechargeable batteries at the Center for Functional Nanomaterials (CFN). The CFN is a Department of Energy Office of Science user facility. The batteries had anodes (the positively charged side of a battery) made of magnesium (Mg) metal. Scientists used scanning transmission electron microscopy (STEM), which can take images of extremely small objects, to measure the batteries as they operated. The study revealed a combination of nanometer-scale features that were key to improving the battery’s performance. In particular, they found it was important for extremely tiny crystals of Mg to form on the anode as the battery worked.
The Impact
Magnesium anodes can store five times as much energy in the same space as graphite anodes. Commercial lithium-ion batteries currently use graphite anodes. Magnesium anodes also have a unique operating mechanism. This mechanism enables batteries to charge and discharge quickly at freezing temperatures (0 °C). This characteristic is important for using these types of batteries in cars and other transportation applications.
Summary
The morphological control of electrochemically deposited metallic anodes, such as Li, Zn, and Mg, under high applied rates is essential for the development of high-energy-density batteries. For transportation applications, maximizing high rates and high energy density is key to attaining viable customer acceleration and range expectations, respectively. In this work, the in-situ generation of Mg nanocrystals allowed cycling under high rates (10 mA cm–2) and reduced temperature (0 °C) for the very first time. Through operando STEM analysis, we discovered a highly functional solid electrolyte interphase (SEI), a first of its kind, which enabled continuous deposition and dissolution of Mg without internal shorting. The unique morphology of the deposited Mg and the functional capability of the SEI are key to future development of practical metallic Mg anodes.
Contact
Kim Kisslinger
Center for Functional Nanomaterials, Brookhaven National Laboratory
kisslinger@bnl.gov; 631-344-5147
Funding
Research carried out at the Center for Functional Nanomaterials, which is supported by the U.S. Department of Energy, Office of Science, Office of Basic Energy Sciences.
Publications
N. Singh, T. S. Arthur, O. Tutusaus, J. Li, K. Kisslinger, H. L. Xin, E. A. Stach, X. Fan, R. Mohtadi, “Achieving High Cycling Rates via In Situ Generation of Active Nanocomposite Metal Anodes,” ACS Applied Energy Materials 1, 4651 (2018). [DOI: 10.1021/acsaem.8b00794]

