Efficient adsorption-based gas separation requires maximizing both the adsorbent’s selectivity for the gas of interest and its recyclability
November 24, 2017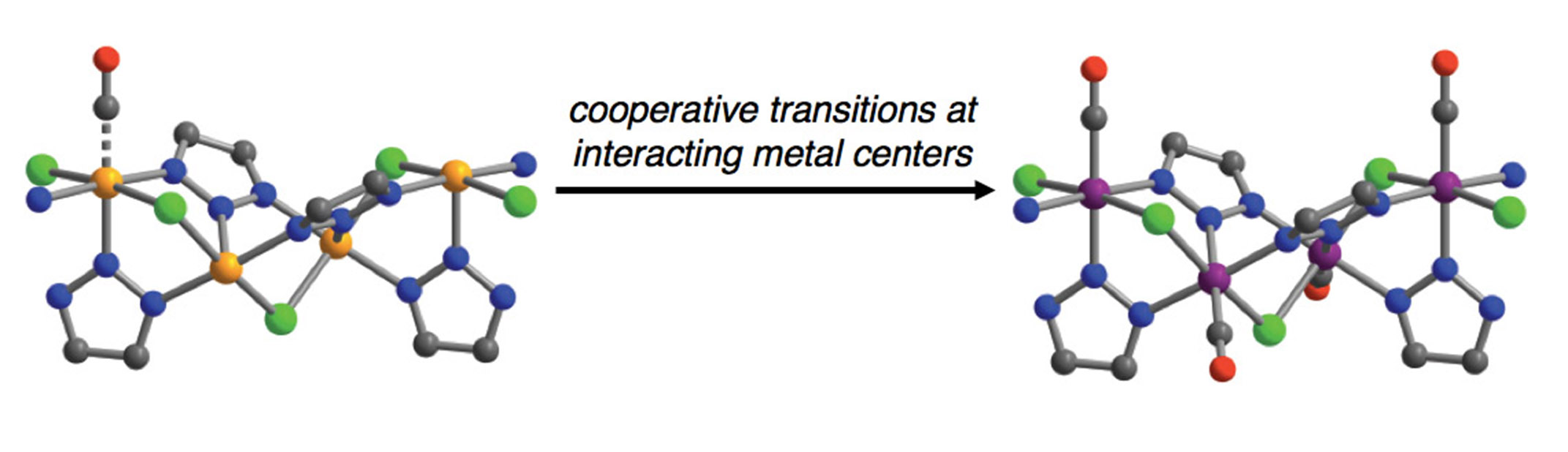
The Science
Inspired by hemoglobin's cooperative mechanism for binding and releasing oxygen, scientists designed new metal-organic frameworks (MOFs) that cooperatively collect and release carbon monoxide (CO) with very little energy input. Each framework has a chain of iron centers along a porous network. In these materials, CO binding at one iron site alters the electronic state of neighboring iron sites in a coordinated way. The coordination leads to the adsorption of the remaining CO along the chain in a zipper-like fashion. Similarly, with a small increase in temperature, a reversal of iron's electronic states occurs as the CO is cooperatively released from these materials.
The Impact
The new mechanism for CO adsorption in these systems allows for significantly higher efficiencies than traditional adsorbents by combining high separation capacities with small temperature swings. These adsorbents are immediately applicable in CO separation processes such as high volume steel manufacturing and syngas production. The approach is broadly applicable to create new adsorbents for the separation of other industrially relevant gases.
Summary
Efficient adsorption-based gas separation requires maximizing both the adsorbent's selectivity for the gas of interest and its recyclability — its easy regeneration under mild conditions. The use of communicating and responsive metal centers to enable cooperative adsorption can be used as a broader design principle for new MOFs for highly efficient separations of industrially relevant gases. Researchers at the Center for Gas Separations Relevant to Clean Energy Technologies (CGS), a DOE Energy Frontier Research Center, have now designed and demonstrated just such an efficient mechanism for CO separation in MOFs. Specifically, these materials contain a chain of interacting iron sites along the cylindrical channels where CO can adsorb. Above a threshold CO pressure, CO binding at an iron site triggers a transformation of electronic configuration more favorable for CO adsorption at neighboring iron sites, leading to a coordinated adsorption reaction. Similarly, with a small increase in temperature, a reversal of iron's electronic states occurs as the CO molecules cooperatively desorb along the chain of iron sites in these materials. Due to this adsorption/desorption mechanism, these MOFs can exhibit large working capacities utilizing small temperature swings, making them highly energy efficient in terms of regeneration while also remaining selective for CO adsorption. Importantly, the research shows that the electronic spin transition is highly tunable through variation of the organic linkers of the framework. Hence, it is possible to design next generation materials with further improvements for CO separation from different gas mixtures. Yet another transformational aspect of this research is that a similar approach can be used to design new MOFs for the separation of a variety of other industrially relevant gases such as acetylene, ethylene, propylene, and dinitrogen.
Contact
Jeffrey R. Long
University of California, Berkeley, Professor of Chemistry and Professor of Chemical and Biomolecular Engineering
jrlong@berkeley.edu
Funding
This research was supported through the Center for Gas Separations Relevant to Clean Energy Technologies, an Energy Frontier Research Center (EFRC) funded by the U.S. Department of Energy (DOE), Office of Science, Office of Basic Energy Sciences under award DE-SC0001015. Powder X-ray diffraction data were collected at Beamline 11-BM and Beamline 17-BM at the Advanced Photon Source, a DOE Office of Science user facility, operated by Argonne National Laboratory under contract DE-AC02-06CH11357.
Publications
D.A. Reed, B.K. Keitz, J. Oktawiec, J.A. Mason, T. Runcevski, D.J. Xiao, L.E. Darago, V. Crocella, S. Bordiga, and J.R. Long, "A spin transition mechanism for cooperative adsorption in metal-organic frameworks." Nature 550, 96-100 (2017). [DOI: 10.1038/nature23674]
Related Links
Lawrence Berkeley National Laboratory news release: Hints from hemoglobin lead to better carbon monoxide storage
Highlight Categories
Performer/Facility: University, SC User Facilities, BES User Facilities, APS

