Second-generation bioethanol is derived from cellulose-based feedstocks. Production of such bioethanol is expected to grow significantly worldwide
January 26, 2017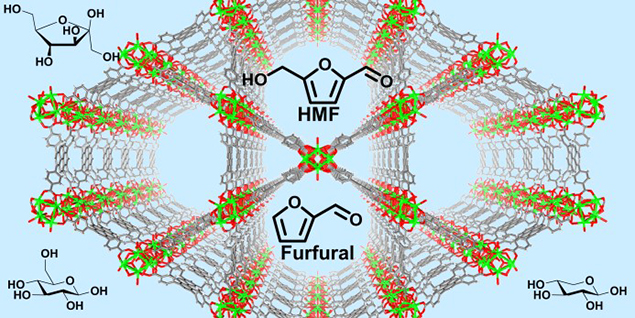
The Science
In creating fuels from the woody materials of plants – the most abundant source of biomass on earth – scientists must separate the desirable sugars from byproduct chemicals including toxins. A team from the University of California at Berkeley, Northwestern University, and Hokkaido University discovered a honeycomb-like structure called NU-1000 that selectively adsorbs toxic furanics, while completely excluding desirable sugars from the same watery mixture. The NU-1000 even works when the sugar is present in several-hundred-fold excess.
The Impact
This research may enable scientists to change how biomass is prepared for conversion into fuels and chemicals. Using NU-1000 allows the potential of greater productivity and efficiency. Such improvements, when producing biomass-derived products, could bring costs down and reduce carbon dioxide emissions.
Summary
Second-generation bioethanol is derived from cellulose-based feedstocks. Production of such bioethanol is expected to grow significantly worldwide beyond its 2014 volume, which was already at 340 million liters per year. During production of second-generation biofuel, as well as sugar-derived chemicals, a major limitation of the required pretreatment of lignocellulosic biomass is toxicity to enzymes and fermentation organisms. However, the results from a group of researchers led by Alexander Katz of the University of California at Berkeley represent an important advance. The team's results could lead to the development of enzyme-like selective adsorbent materials for removing aromatic toxins such as furanics from aqueous sugar mixtures destined for fermentation. The crucial advance relies in the crystallinity and organization of organic functional groups in the metal-organic framework (MOF), NU-1000, to achieve this complete separation. The results of this research may enable development of new pretreatment methodologies for biomass-derived products. Existing treatments were often deemed too harsh because they resulted in sugar mixtures that exhibited unacceptably high toxicity to enzymes and fermentation organisms. However, scientists can use NU-1000 to modify existing pretreatment approaches. In these cases, NU-1000 may enable harsher pretreatment than before (that is, longer duration and/or higher temperatures) for higher sugar, fuel, and chemical yields. Ultimately, NU-1000 may result in substantial reductions in carbon dioxide emissions and cost savings.
Contact
Alexander Katz
University of California at Berkeley, Department of Chemical and Biomolecular Engineering
askatz@berkeley.edu
Funding
Funding for this work was provided by the U.S. Department of Energy, Office of Science, Office of Basic Energy Sciences, Division of Chemical Sciences, Geosciences and Biosciences (award DE-FG02-05ER15696). O.K.F. gratefully acknowledges support from the Inorganometallic Catalyst Design Center, an Energy Frontier Research Center, funded by the U.S. Department of Energy, Office of Science, Basic Energy Sciences, under award DESC0012702. M.Y. received Grant-in-Aid for postdoctoral fellows from Japan Society for the Promotion of Science (JSPS, 14J01171).
Publications
M. Yabushita, P. Li, H. Kobayashi, A. Fukuoka, O.K. Farha, and A. Katz, "Complete furanics-sugar separations with metal-organic framework NU-1000." Chemical Communications 52, 11791-11794 (2016). [DOI: 10.1039/C6CC05864G]
M. Yabushita, P. Li, V. Bernales, H. Kobayashi, A. Fukuoka, L. Gagliardi, O.K. Farha, and A. Katz, "Unprecedented selectivity in molecular recognition of carbohydrates by a metal-organic framework." Chemical Communications 52, 7094-7097 (2016). [DOI: 10.1039/C6CC03266D]
Related Links
Chemical & Engineering News article: MOF Traps Molecules that Gum Up Bioethanol Production
Highlight Categories
Performer/Facility: University
Additional: Collaborations, International Collaboration

