Molecular cage to trap chloride could help reduce rising level of salt contaminants flowing into freshwater streams & lakes across the United States.
November 18, 2019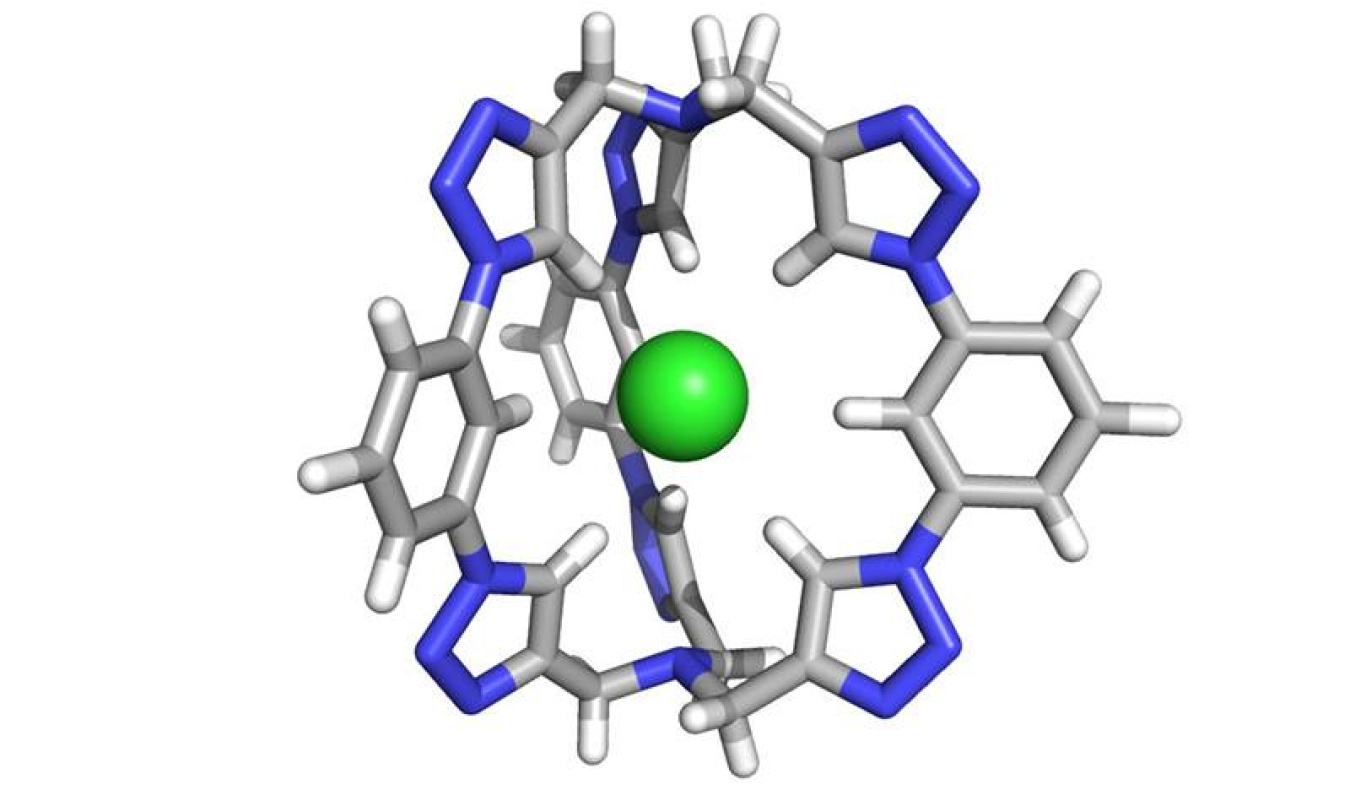
The Science
Researchers devised a powerful new molecule for the extraction of salt from liquid. The molecule captures chloride. Built using chemical bonds previously regarded as too weak, the new molecule is highly effective. It binds chloride using carbon-hydrogen bonds, previously regarded as too weak to create stable interactions with chloride. The new salt-extraction molecule is composed of six triazole motifs, which together form a 3-D cage perfectly shaped to trap chloride.
The Impact
Salts seeping into freshwater streams and lakes reduce access to drinkable water across the globe. Salts come from oil extraction, road salts, water softeners, and the natural weathering of rock. It takes just one teaspoon of salt to permanently pollute five gallons of water. The research and the resulting molecular cage could help increase the amount of drinkable water on Earth.
Summary
About 272 metric tons of dissolved solids, including salts, enter U.S. freshwater streams per year, according to estimates by the U.S. Geological Survey. These solids pollute the water, making it undrinkable. The challenge is to remove the salts quickly and efficiently. Many of these salts contain chloride ions. Researchers have looked at flexible molecular cages to capture the chloride. Now, researchers have designed a rigid molecular cage that draws in chloride ions. The rigidity also enables the molecule to retain its shape after the central chloride has been lost, compared to other designs that collapse under the same circumstances due to their flexibility. This gives the molecule greater efficacy and versatility. The team made the molecular cage using carbon-hydrogen bonds, which are weaker than typical nitrogen-hydrogen bonds. Compared to cages made with stronger bonds, the new cage performs several orders of magnitude better. This rigid molecular cage holds promise for water purification and potentially other applications.
Contact
Amar H. Flood
Indiana University
aflood@indiana.edu; (812) 856-3642
Funding
The Department of Energy, Office of Science, Office of Basic Energy Sciences, Chemical Sciences, Geosciences, and Biosciences division funded this research. A.H.F. acknowledges support from the Waterman Professorship.
Publications
Y. Liu, W. Zhao, C.H. Chen, and A.H. Flood, “Chloride capture using a C–H hydrogen bonding cage.” Science 23, eaaw5145 (2019). [DOI: 10.1126/science.aaw5145]
Related Links
Indiana University Bloomington news release: Building a better salt trap: IU researchers synthesize a molecular 'cage' to trap chloride

