Theorists show how a new quantum device could control a chemical reaction remotely, changing our understanding of how reactions can work.
April 18, 2019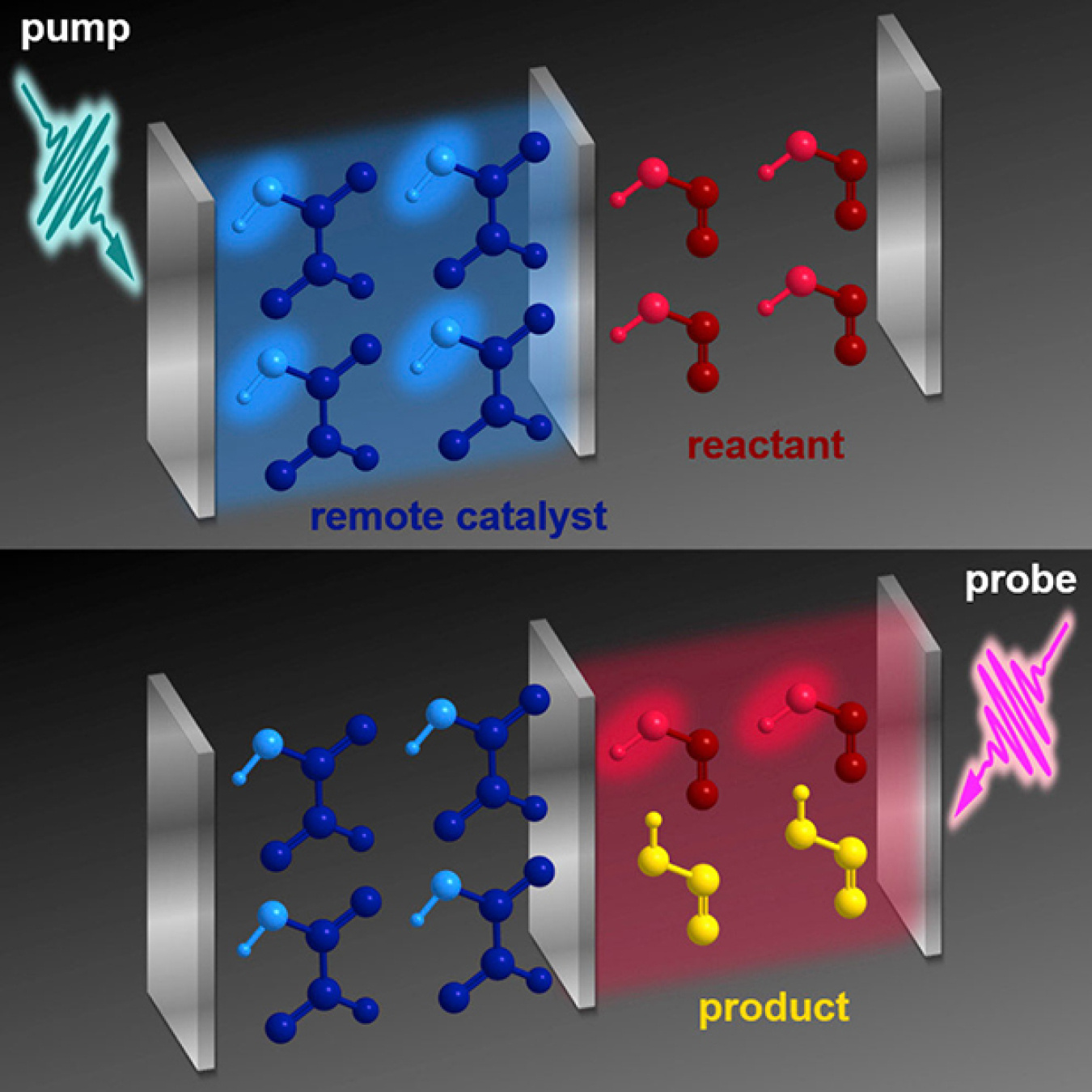
The Science
Students learn in high school that molecules must be in contact to react chemically. But what if that's not always true? It's that idea, which challenges textbook "laws," a team of theorists explored. They showed that even though it is in a completely different container from reactants, a catalyst could make a reaction happen. That is, a catalyst caused nitrous acid to change shape without touching it. The team's theory challenges conventional wisdom about what it takes to make a reaction happen.
The Impact
High school textbooks state that molecules need to touch to react. In this theoretical study, scientists designed a quantum device that separates the catalyst from the starting chemicals. Using light, the scientists excited the catalyst to control the adjacent reaction. The setup could let chemists reconfigure chemical bonds that they can't access in other ways.
Summary
Some chemical bonds are tough to re-arrange because it's hard to access them. This aligns with the conventional wisdom that to make and break bonds, a catalyst must be in physical contact with the bond. Researchers challenged that wisdom. The team showed how it's possible to alter bonds when the catalyst and reactants are separate, by exploiting a strong coupling between light and matter that can lead to changes in chemical reactions. The team proposed a quantum device where a mirror separates a catalyst (glyoxylic acid) from the reactant (cis-nitrous acid). They excited the catalyst molecules and their container (an optical "cavity") using a short bust of light from an ultrafast laser, to form polaritons, quasi-particles made of both light and matter. The polaritons boost the cis-trans isomerization of nitrous acid by an order of magnitude. Forming polaritons causes the molecules in the containers to behave like a big supermolecule. If you affect one part of the supermolecule, you affect the other. The team believes experimentalists will soon be able to build the quantum device and show the remote control of chemistry in the laboratory soon.
Contact
Joel Yuen-Zhou
University of California, San Diego
joelyuen@ucsd.edu
Funding
The University of California, San Diego Chancellor's Research Innovation Scholarships postdoctoral award (R.F.R. carried out exploratory studies of the presented remote control in the linear regime), Air Force Office of Scientific Research (coarse-grained the model of IVR from the literature), and Department of Energy, Office of Science, Early Career Research Program (M.D. and J.Y.Z. for development of the remote-control model in the nonlinear regime via input-output theory) funded this work.
Publications
M. Du, R.F. Ribeiro, and J. Yuen-Zhou, "Remote control of optical cavities." Chem (2019). [DOI: 10.1016/j.chempr.2019.02.009]
Related Links
University of California, San Diego news release: Scientists 'game' for remote-control chemistry of the future
Chemistry World article: Theory suggesting a catalyst never needs to touch its reactant breaks textbook principle

