Metal-organic frameworks designed with a topology-guided approach show higher efficiency than commercial benchmarks.
June 13, 2019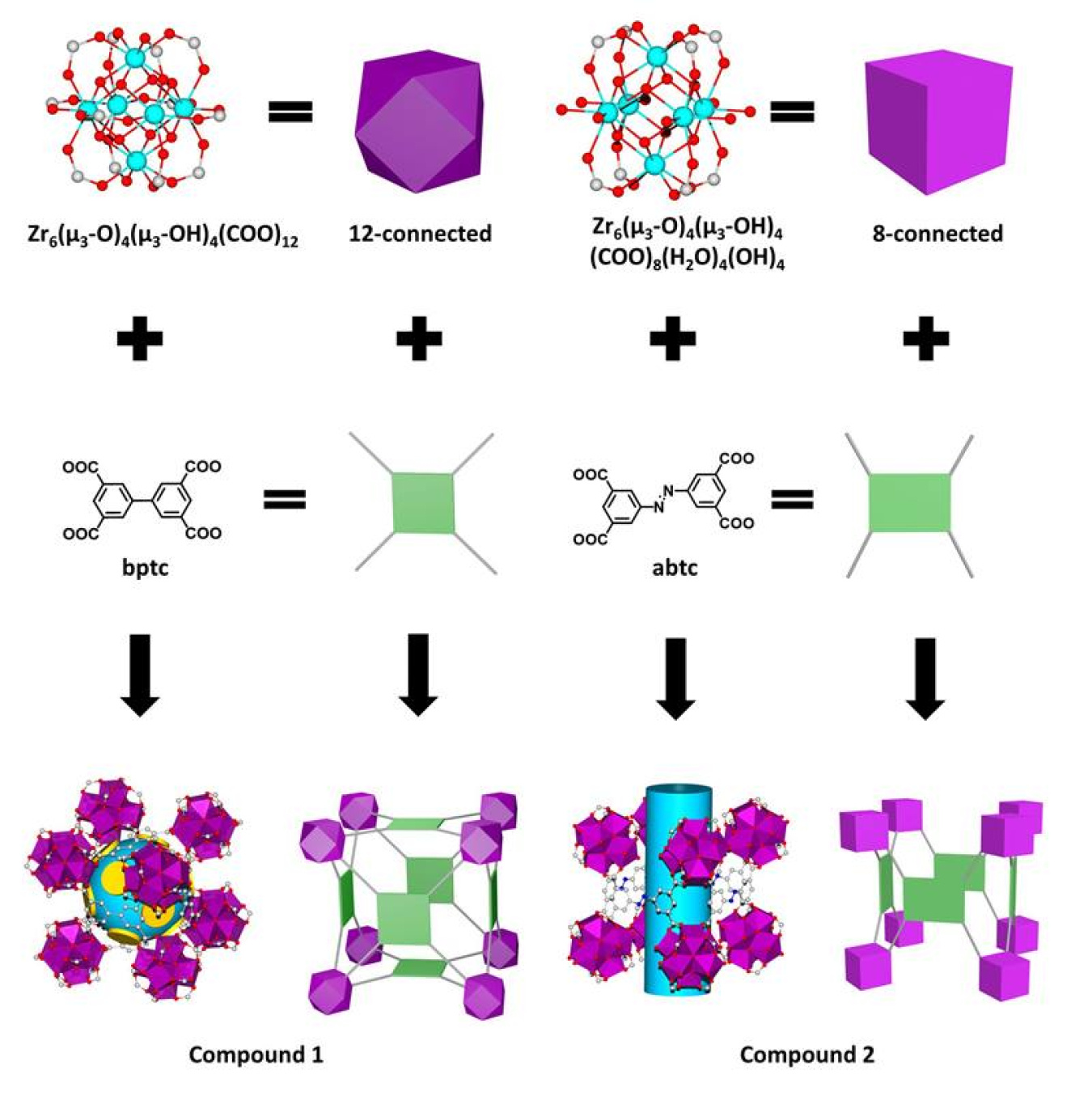
The Science
Separating desirable chemicals from others can use a lot of energy. Certain porous materials can separate molecular mixtures using different mechanisms. In this study, scientists designed two porous materials, zirconium-based metal-organic frameworks (Zr-MOFs), to separate hydrocarbons containing six carbon atoms. This separation is a key step in fuel production. One of the Zr-MOFs used preferential adsorption to pick out a straight hydrocarbon chain from a mixture of different structures. The uptake was ~70% higher than a common benchmark. The other new material worked like a sieve. It quickly separated different structures with an efficiency not possible with a common benchmark material.
The Impact
The cost-effective separation of alkanes is of paramount importance in the petroleum industry. Alkanes are chemicals with specific arrangements of hydrogen and carbon atoms. Such separations are needed to produce high-octane gasoline. Industry experts must separate alkanes with the same number of linked carbons but with different structures and different properties (isomers). Some of the materials designed and synthesized in this study efficiently separate the isomers. Therefore, these materials hold strong promise for use in alkane separation processes such as fuel refining.
Summary
As an alternative to conventional energy-intensive distillation methods, separations using porous materials offers lower energy cost and higher efficiency. This study demonstrates the design and synthesis of a series of Zr-MOFs with optimized pore structure for efficient separation of alkane isomers containing six carbons (C6). Researchers conducted breakthrough experiments that offer a quantitative measure of the capability of these materials to separate C6 alkane isomers. The team controlled the pore shape and size by altering the size and geometry of the organic component of the frameworks. Their changes had a major impact on isomer separation efficiency. One material with cage-like pores, (Zr6O4(OH)4(bptc)3), adsorbs a large amount of a linear C6 isomer (n-hexane); the structure of the cage-like pores excluded all branched C6 isomers. The n-hexane uptake is ~70% higher than that of a benchmark adsorbent, zeolite-5A. Another Zr-MOF, (Zr6O4(OH)8(H2O)4(abtc)2), with channel-like pores discriminated between all three types of C6 isomers, yielding a high separation factor for a C6 alkane with one carbon branch (3-methylpentane) over a C6 alkane with two carbon branches (2,3-dimethylbutane). This level of separation efficiency exceeds that of other MOFs and is not possible using commercial zeolite-5A. A detailed structural analysis reveals the unique topology, connectivity, and relationship of these compounds and demonstrates the ability to design functional materials for energy-relevant applications.
Contact
Jing Li
Rutgers University
jingli@rutgers.edu
Funding
This study was funded by the following organizations: Department of Energy (DOE), Office of Science, Materials Sciences and Engineering Division; the King Abdullah University of Science and Technology Competitive Center Funding (Y.H.); Simons Foundation endowed sabbatical (T.T.); Russian Government (D.M.P.); and the Russian Science Foundation (E.V.A.). The team used resources at the Advanced Light Source, a DOE Office of Science user facility.
Publications
H. Wang, X. Dong, J. Lin, et al., “Topologically guided tuning of Zr-MOF pore structures for highly selective separation of C6 alkane isomers.” Nature Communications 9, 1745 (2018). [DOI: 10.1038/s41467-018-04152-5]

