The development of artificial photosynthetic systems for making chemical fuels from solar energy is a long-term research objective that spans many
April 30, 2016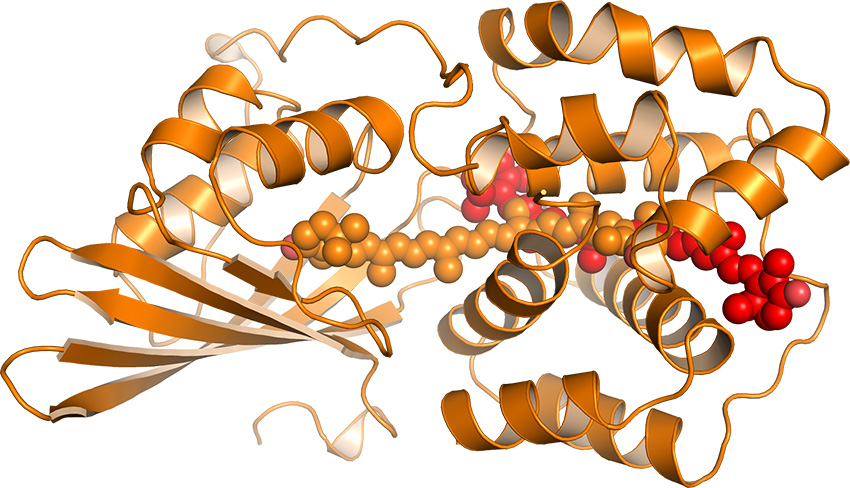
The Science
Too much sunlight can harm the ability of plants to turn the sun's energy into fuels; nature has solved the problem by dissipating the unwanted energy as heat. With an eye to learning from nature's success, scientists characterized the orange-colored protein that protects cyanobacteria from overexposure to sunlight. They found that the orange-ish protein shifts to a reddish color that helps dissipate excess energy harmlessly as heat.
The Impact
The findings could be useful for design of artificial photosynthetic systems, which use intermittent sunlight to create use-any-time fuels.
Summary
The development of artificial photosynthetic systems for making chemical fuels from solar energy is a long-term research objective that spans many scientific and technical challenges. One key question is the ability of the photosynthetic system to protect itself from photodamage. Natural sunlight is damaging to the photosynthetic systems of plants, algae, and cyanobacteria, and is expected to be similarly harmful to highly efficient artificial systems. Nature has developed a mechanism for photoprotection that involves removing excess energy from the system harmlessly as heat; however, no such process currently exists for artificial systems. In an effort to understand mechanisms of natural photoprotection, researchers at Lawrence Berkeley National Laboratory and Michigan State University have characterized the orange carotenoid protein (OCP) that provides photoprotection in cyanobacteria. Upon exposure to sunlight, the OCP is photoactivated and converts from an "orange, light-absorbing" state to a "red, photoprotection" state that binds to the cyanobacterial antenna to dissipate heat. Using x-ray crystallography and x-ray footprinting, this work has shown that conversion from the orange to red state is induced by a 12 Å shift of a carotenoid pigment within the OCP. The pigment shift creates different carotenoid-protein interactions that cause the protein to change shape, while also providing the ability for the newly formed "red" state protein to bind to the cyanobacterial antenna and dissipate heat. Similar carotenoid motions may occur in plants and algae, which also rely on carotenoid pigments bound to proteins for photoprotection.
Contact
Cheryl Kerfeld
Lawrence Berkeley National Laboratory/Michigan State University
ckerfeld@lbl.gov, kerfeldc@msu.edu
Funding
Supported by the U.S. Department of Energy (DOE), Office of Science, Basic Energy Sciences, Chemical Sciences, Geosciences, and Biosciences Division award DE-FG02-91ER20021. The Advanced Light Source at Lawrence Berkeley National Laboratory is supported by the Office of Science, Office of Basic Energy Sciences, DOE under contract no. DE-AC02-05CH11231. This research used resources of the National Energy Research Scientific Computing Center, a DOE Office of Science User Facility supported by the DOE Office of Science under contract no. DE-AC02- 05CH11231 and the Joint BioEnergy Institute supported by the DOE Office of Science, Office of Biological and Environmental Research under contract DE-AC02-05CH11231. A.W., A.T., C.B., and D.K. are supported by a grant from the Agence Nationale de la Recherche (ANR, project CYANOPROTECT), and used resources of CNRS and the Commissariat à l'Energie Atomique (CEA).
Publications
R. L. Leverenz, M. Sutter, A. Wilson, S. Gupta, A. Thurotte, C. Bourcier de Carbon, C. J. Petzold, C. Ralston, F. Perreau, D. Kirilovsky, and C. A. Kerfeld "A 12 Å Carotenoid Translocation in a Photoswitch Associated with Cyanobacterial Photoprotection ." Science, 348, 1463 (2015). [DOI: 10.1126/science.aaa7234]
." Science, 348, 1463 (2015). [DOI: 10.1126/science.aaa7234]
Related Links
Lawrence Berkeley National Laboratory Press Release
Highlight Categories
Performer/Facility: SC User Facilities, ASCR User Facilities, NERSC, BES User Facilities, ALS
Additional: Collaborations, International Collaboration

