Studies pinpoint the active site of a catalyst that converts sunlight to liquid fuels.
July 15, 2020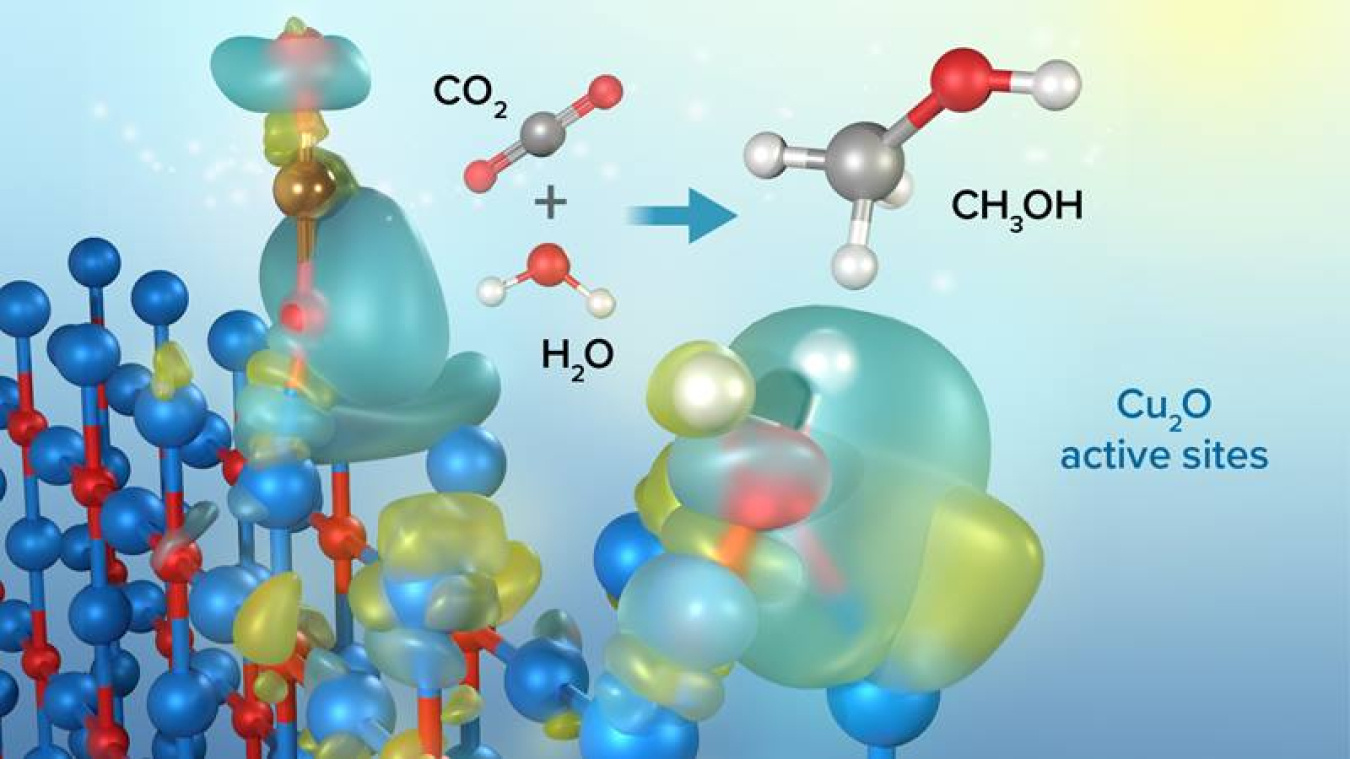
The Science
Scientists have revealed the exact structure of a catalyst that transforms carbon dioxide (CO2) and water into liquid fuel in the presence of light. Catalysts are chemicals that accelerate reactions. The researchers used advanced analytical techniques and computer models to study a specific promising catalyst, Copper(I) oxide (Cu2O).
The Impact
The efficient conversion of CO2 into liquid fuel could help to reduce our dependence on fossil fuels. Chemical conversion of CO2 using sunlight offers a promising way to store solar energy in the form of liquid chemical fuels. These catalysts that use light are called photocatalysts. Specific catalysts can help facilitate these conversions without unnecessary energy loss. Identifying the active part of catalyst structures, the subject of this study, is an important step in the design of photocatalysts. This ability will help researchers create photocatalysts for converting CO2 into fuels such as methanol and ethylene and for other energy applications.
Summary
Photoreduction of CO2 is a process that uses sunlight to efficiently store solar energy in the form of chemical fuels. This study reveals the detailed structure of a catalyst called a photocatalyst used in this process. This photocatalyst helps convert CO2 into stored solar energy in the form of methanol, a liquid chemical fuel. In this study, scientists used a combination of advanced analytical techniques and computer models to identify the specific active sites, or areas on the photocatalyst surface that target specific parts of the CO2 molecules during the chemical process of converting CO2 into fuel. The researchers used the photocatalyst Copper(I) oxide (Cu2O) because of its unique electronic properties and because it is made of commonly available materials, copper and oxygen. Cu2O also absorbs visible light at frequencies covering most of the visible solar spectrum.
Cu2O is very efficient at converting CO2 to liquid fuel methanol as the sole product. The internal quantum yield for unassisted wireless photocatalytic reduction of CO2 to methanol using Cu2O crystals was approximately 72 percent. They expect that revealing the active site of Cu2O will inspire the development of new classes of catalyst materials with improved activity.
The researchers used X-ray microscopy at the Advanced Photon Source and transmission electron microscopy at the Center for Nanoscale Materials (CNM), two Department of Energy Office of Science user facilities. Further analysis at CNM used multimodal correlated characterization of a single Cu2O particle, ex situ ensemble measurements, and density functional theory calculations. By demonstrating the ability to multi-modally identify the catalytically active sites on the individual Cu2O crystal and to observe them in action, the research is a significant advance that could enable the design of future high-performance CO2 reduction photocatalysts.
Contact
Tijana Rajh and Yuzi Liu
Center for Nanoscale Materials, Argonne National Laboratory
rajh@anl.gov and yuziliu@anl.gov
Funding
This work was performed at the Center for Nanoscale Materials and the Advanced Photon Source at Argonne National Laboratory, both Department of Energy (DOE) Office of Science user facilities. Partial support was provided by Argonne’s Laboratory Directed Research and Development program. The computational studies and X-ray photoemission spectroscopy were supported by the DOE Office of Science, Office of Basic Energy Sciences, Division of Materials Sciences and Engineering. Three of the researchers acknowledge grants of computer time through the LCRC Blues Cluster at Argonne. One of the researchers acknowledges financial support by the DOE Office of Science, Office of Basic Energy Sciences, Division of Chemical Sciences, Geosciences, and Biosciences.
Publications
Wu, Y.A. et al., “Facet-dependent active sites of a single Cu2O particle photocatalyst for CO2 reduction to methanol,” Nature Energy 4, 957-968 (2019). [DOI: 10.1038/s41560-019-0490-3]
Related Links
Argonne press release: Scientists devise catalyst that uses light to turn carbon dioxide to fuel

