Ozone injection may lead to cleaner, more efficient internal combustion engines.
February 16, 2022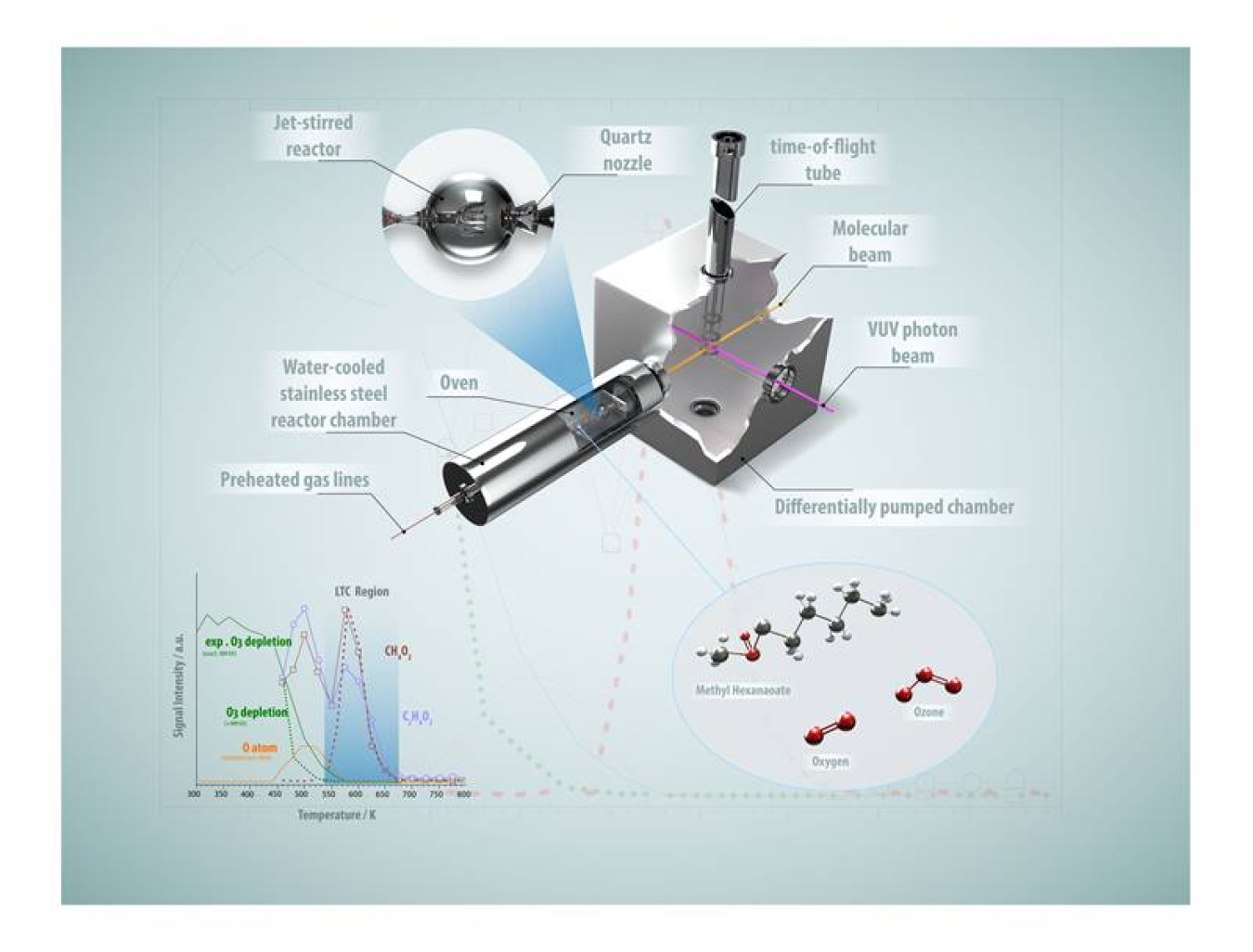
The Science
Low temperature chemistry (LTC) is a special set of reactions that take place at what chemists consider relatively low temperatures: roughly 400-700 Kelvin (260-800 degrees F). Studying LTC is important to the basic science of chemistry. It is also useful for understanding how internal combustion engines burn fuel. In these engines, the ignition of fuel is surprisingly complex, occurring in two separate steps. This two-step process affects how engines produce harmful emissions. One way to make an LTC reaction faster is to inject ozone into a system. This study examined the use of ozone injection with methyl hexanoate (MHX). Researchers use this substance to study the chemistry of the main components of biodiesel. The study found new ways that combustion can operate at temperatures at the very low end of the LTC range.
The Impact
This research discovered a new combustion process for MHX. It occurs at relatively low temperatures (approximately 440 Kelvin, or 330 degrees F) when small amounts of ozone are added to a mix of MHX and oxygen. This finding can help reduce emissions of soot and nitrogen oxide from internal combustion engines. The results are relevant to both biodiesel and conventional fuel engines. The findings also offer new insights on the chemistry of oxidation. They will also contribute to research on atmospheric chemistry.
Summary
The researchers investigated the reactivity of methyl hexanoate/oxygen mixtures over a temperature range (460-940 Kelvin) in the presence of small amounts of ozone in an externally heated, atmospheric pressure jet-stirred reactor. The researchers identified a previously undetected oxidation regime at temperatures below the typical low-temperature regime (extreme low-temperature combustion or ELTC). The researchers identified key intermediate species using a mass spectrometer and ultraviolet radiation generated by the Chemical Dynamics Beamline of the Advanced Light Source, a Department of Energy (DOE) user facility. The experimental data indicates that the chemistry in the ELTC regime is initiated by thermal decomposition of ozone, followed by reactions from methyl hexanoate by oxygen atoms from the ozone.
There have been many investigations into the oxidation of unsaturated hydrocarbons by ozone. However, investigations into the interactions with molecules without a C=C double bond have been scarce. The observation in this research of a new ELTC regime addresses the societal need for clean combustion, as the addition of ozone enables sustainable oxidation near 500 Kelvin, thus eliminating emission of toxic byproducts such as nitrogen oxide and soot. Furthermore, this work provides atmospheric chemistry researchers with information on the formation pathways of highly oxygenated species that are considered to form secondary organic aerosols, another pollutant. Because ozone is a long-lived air-plasma intermediate, this research also improves understanding of plasma-initiated oxidation processes.
Contact
Nils Hansen
Sandia National Laboratories
Email: nhansen@sandia.gov
Funding
This work was supported by the DOE Office of Science, Basic Energy Sciences, Chemical Sciences, Geosciences and Biosciences Division, as part of the Gas Phase Chemical Physics Program.
Publications
Rousso, A. C., Jasper, A. W., Ju, Y., and Hansen, N., Extreme Low-Temperature Combustion Chemistry: Ozone-Initiated Oxidation of Methyl Hexanoate. Journal of Physical Chemistry A, 124, 9897-9914 (2020). [DOI: 10.1021/acs.jpca.0c07584]

