Mercury is a toxic element that occurs naturally and as an anthropogenic pollutant in the environment. The neurotoxin monomethylmercury (MMHg) is a
Biological and Environmental Research
December 18, 2018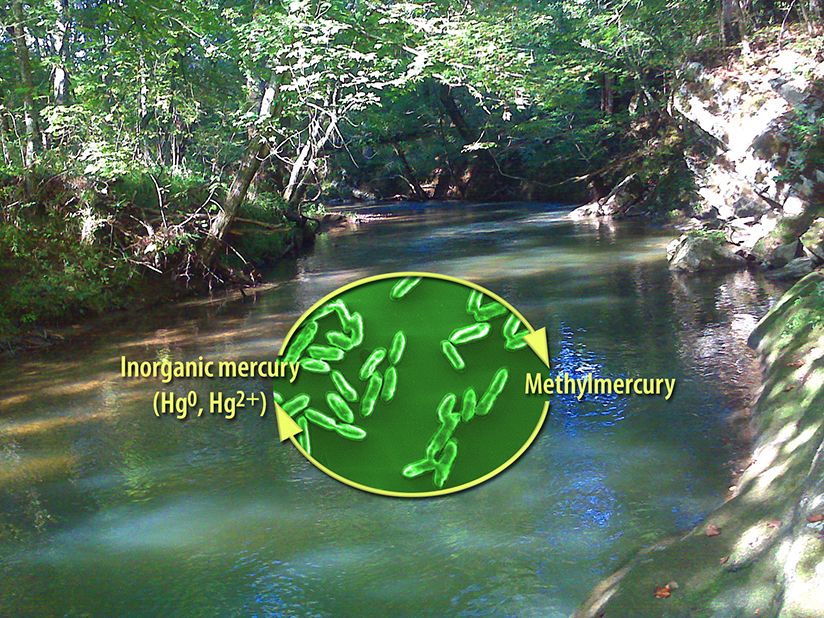
The Science
While volcanoes and forest fires release mercury, they are relatively small sources compared to the combustion of coal, oil, and other fuels. Mercury is toxic. Microbes turn mercury into a neurotoxin called methylmercury. They also turn the neurotoxin back into inorganic mercury. To predict the levels of inorganic mercury and methylmercury in the environment, scientists need to know how fast microbes act. Using new experiments and re-analyses of previous experiments, scientists developed a model that describes the production of methylmercury over time. The new model takes into account competing processes and results in faster rates of production than previously estimated.
The Impact
With the new model, scientists can simulate methylmercury production and transport. The model shows that methylmercury production is likely much larger than current estimates. Why does this matter? Methylmercury has adverse effects on young children and developing embryos. Better predicting mercury levels helps experts protect people and the environment.
Summary
Mercury is a toxic element that occurs naturally and as an anthropogenic pollutant in the environment. The neurotoxin monomethylmercury (MMHg) is a particular concern because it biomagnifies in aquatic environments and has adverse development effects on young children and developing embryos. MMHg is formed in the environment from inorganic mercury through the action of microorganisms in a process called mercury methylation. Because of its toxicity, scientists have attempted, numerous times, to measure mercury methylation and MMHg demethylation rates in various environmental settings with differing results. Even in laboratory experiments, rates for the methylation of mercury to MMHg often exhibit kinetics that are inconsistent with first-order kinetic models. In a new study, scientists from Oak Ridge National Laboratory used time-resolved measurements of filter-passing mercury and MMHg during methylation/demethylation assays, and they re-analyzed previous assays. Then they used a multi-site kinetic sorption model to show that competing kinetic sorption reactions can lead to apparent non-first-order kinetics in mercury methylation and MMHg demethylation. The new model can describe the range of behaviors for time-resolved methylation/demethylation data reported in the literature including those that exhibit non-first-order kinetics. Additionally, the team showed that neglecting competing sorption processes could confound analyses of methylation/demethylation assays, resulting in rate constant estimates that are systematically biased low. Simulations of MMHg production and transport in a hypothetical periphyton biofilm bed illustrate the implications of the new model and demonstrate that methylmercury production may be significantly different from projected by single-rate first-order models.
Contact
Program Manager
Paul Bayer
Department of Energy, Office of Science, Biological and Environmental Science
Paul.Bayer@science.doe.gov; (301) 903-5324
Principal Investigator
Scott Brooks
Oak Ridge National Laboratory
brookssc@ornl.gov; (865) 574-6398
Funding
This work was funded by the Department of Energy, Office of Science, Biological and Environmental Research, Subsurface Biogeochemical Program and is a product of the Science Focus Area at Oak Ridge National Laboratory. The isotopes used in this research were supplied by the Department of Energy, Office of Science, Office of Nuclear Physics, Isotope Program.
Publications
T.A. Olsen, K.A. Muller, S.L. Painter, and S.C. Brook, "Kinetics of mercury methylation revisited." Environmental Science & Technology 52(4), 2063(2018). [DOI: 10.1021/acs.est.7b05152]
Related Links
Department of Energy Subsurface Biogeochemical Research Program: Clarifying Rates of Methylmercury Production
Highlight Categories
Performer/Facility: DOE Laboratory

