Insight about energy flow in copper-based material could aid in creating efficient molecular electronics.
April 19, 2019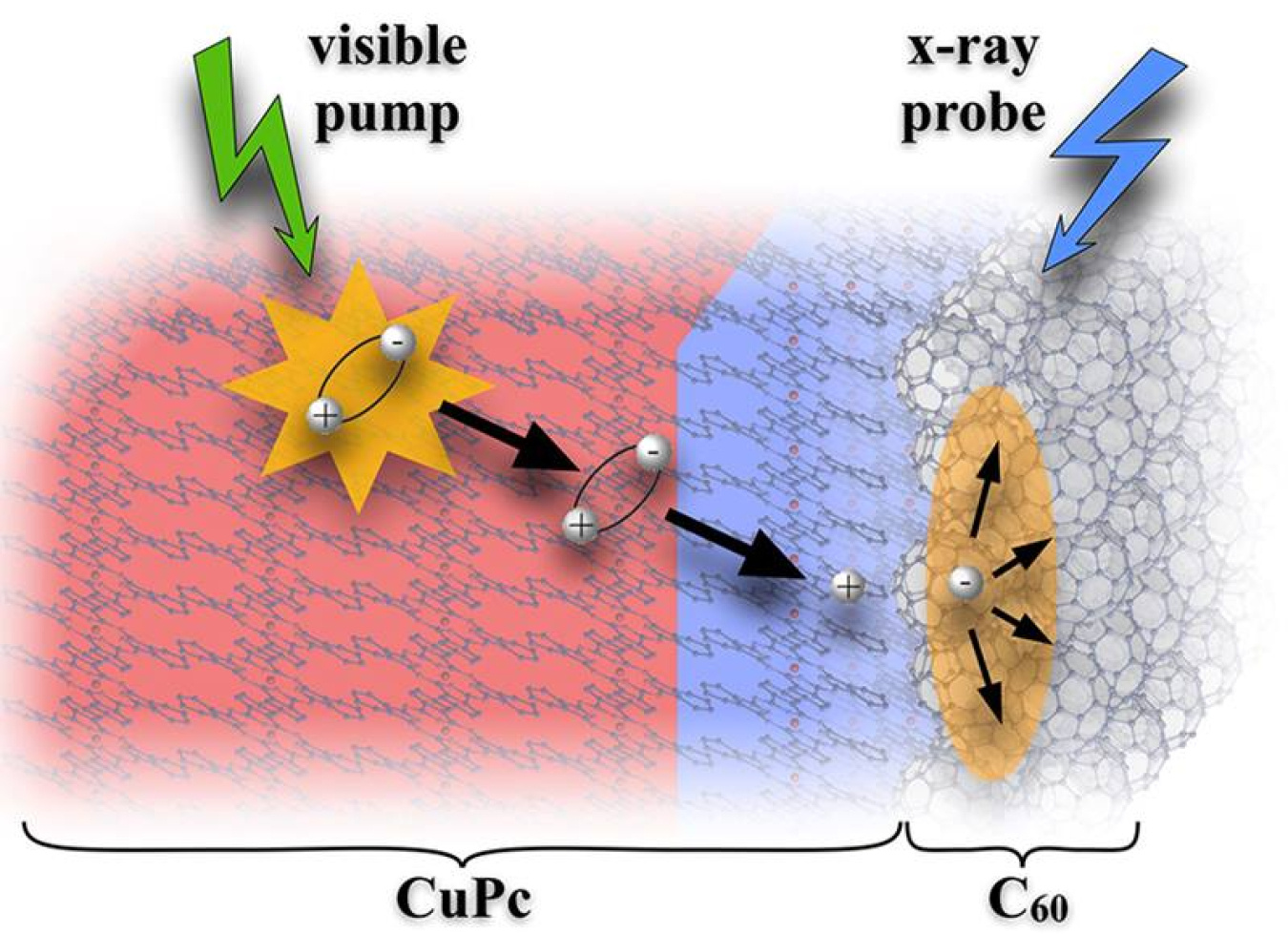
The Science
Next-generation devices based on molecular materials have the potential to efficiently use sunlight to produce electricity or to drive chemical reactions. In contrast to commercial solar cells, where absorption of light directly generates charge that can be extracted as electricity, absorption of light by molecular materials creates uncharged energetic states called excitons. The trick to making an effective device is to efficiently convert the excitons into charge. This study uncovered a new mechanism to create charge from excitons. By following excitons in a model molecular material, a team found that lower energy excitons made deep in the bulk play a larger part in generating charge than previously thought.
The Impact
The team's study shows that a previously neglected, slower pathway creates most of the charge at the interface. This insight shows a pathway that scientists should consider when designing molecular electronics. The study also provides absolute benchmark values for the design of specialized interfaces that efficiently convert light to charge.
Summary
To design molecular solar cells, sensors, etc., researchers need to know how light creates electricity on an atomic scale. This isn't easy because the processes can occur in a trillionth of a second (picosecond). Researchers found a way, using picosecond time-resolved X-ray photoelectron spectroscopy and a model material made of a copper-based electron donor and a carbon acceptor. Specifically, they looked at metal-organic heterojunctions in a copper-phthalocyanine (CuPc) donor and fullerene-based (C60) acceptor. The method lets scientists study how excitons migrate through the material and dissociate into separated charges that are useful for electronic or chemical applications. Their results changed how scientists think about energy in this material. The previous view focused on the higher energy excitons, known as singlet excitons, that are directly generated by the light. Researchers thought the singlet excitons created where the donor and acceptor materials meet generated most of the electricity. Not so. Slower forming, lower-energy triplet excitons that form in the bulk of the copper-based material contribute the majority of charge on a time-averaged basis. The study offers new insights into exciton migration and charge generation in these materials. Also, it provides absolute benchmark values for the design of CuPc-C60 heterojunctions for efficient light-to-charge conversion.
Contact
Oliver Gessner
Lawrence Berkeley National Laboratory
OGessner@lbl.gov
Funding
This work was supported by the "Structure of Matter" program of the Helmholtz Association and by the Director, Office of Science, Office of Basic Energy Sciences, Division of Chemical Sciences, Geosciences, and Biosciences of the Department of Energy (DOE) at the Lawrence Berkeley National Laboratory. O.G. was supported by the DOE Office of Science Early Career Research Program. F.R. acknowledges financial support by the VI 419 of the Helmholtz Association. S.N. acknowledges support by the Alexander von Humboldt Foundation. J.M. acknowledges support from the Advanced Light Source (ALS) Doctoral Fellowship in Residence Program. The ALS is supported by the Director, DOE, Office of Science, Office of Basic Energy Sciences.
Publications
F. Roth, S. Nepply, A. Shavorskiy, T. Arion, J. Mahl, H.O. Seo, H. Bluhm, Z. Hussain, O. Gessner, and W. Eberhardt, "Efficient charge generation from triplet excitons in metal-organic heterojunctions." Physical Review B 99, 020303(R) (2019). [DOI: 10.1103/PhysRevB.99.020303]

