Co-processing biomass-derived oil with crude oil in existing refineries is an effective way to stimulate the market for sustainable transportation fuels. With this approach, the fuel produced by co-producing bio-oil and petroleum can still be “dropped in”
Alternative Fuels and Feedstocks Office
April 21, 2022
Author: Sheila Van Cuyk, Laboratory Relationship Manager for BETO Programs at Los Alamos National Laboratory;
Read Sheila's bio ►
Meet the other bloggers ►
Return to Bioprose blog ►
Upgrading bio-oils with petroleum feedstocks at existing refineries—known as “co-processing”—could offer a fast pathway for lowering the carbon footprint of today’s transportation fuels. With equipment and infrastructure already in place, more than 100 refineries across the country are equipped to integrate bio-oil into their processes. But just where might companies insert bio-oils into existing refinery systems like fluid catalytic crackers and hydrotreaters? If successful, just how much of that renewable or “green” carbon makes it into final fuel products?
In this three-part blog series, the National Renewable Energy Laboratory (NREL), Pacific Northwest National Laboratory (PNNL), and Los Alamos National Laboratory (LANL) offer insights into these pressing co-processing questions—and others—amid the push to dramatically expand the production of climate-friendly fuels. Read part 1 and part 2.
Co-processing biomass-derived oil with crude oil in existing refineries is an effective way to stimulate the market for alternative transportation fuels. With this approach, the fuel produced by co-producing bio-oil and petroleum can still be “dropped in” to existing infrastructure and vehicles.
The challenge, however, lies in quantifying this percentage in order to verify that the fuel product in fact contains renewable carbon. Current verification processes are expensive and unwieldy, so scientists at the U.S. Department of Energy national labs are working on an alternative.
Isotopes: The Signature for Renewable Carbon
The gold standard for assessing the percentage of renewable carbon in co-processed fuel uses accelerator mass spectrometry (AMS) to identify the 14C isotope. This technique works because 14C has a short half-life and no longer exists in the ancient, fossil fuel-derived crude oil, so any instance of it would indicate the presence of bio-oil. Unfortunately, AMS is an expensive process, requiring a large, dedicated facility that cannot easily be incorporated into a refinery.
To address this challenge, a team of researchers at Los Alamos National Laboratory (LANL) led by Zhenghua Li, in collaboration with National Renewable Energy Laboratory and Pacific Northwest National Laboratory, created a solution by developing an accurate, fast, and economic approach for determining renewable carbon content by quantifying the ratio of two other naturally occurring carbon isotopes: 13C/12C.
Varieties of Isotope Hunting: The Difference Is in the Details
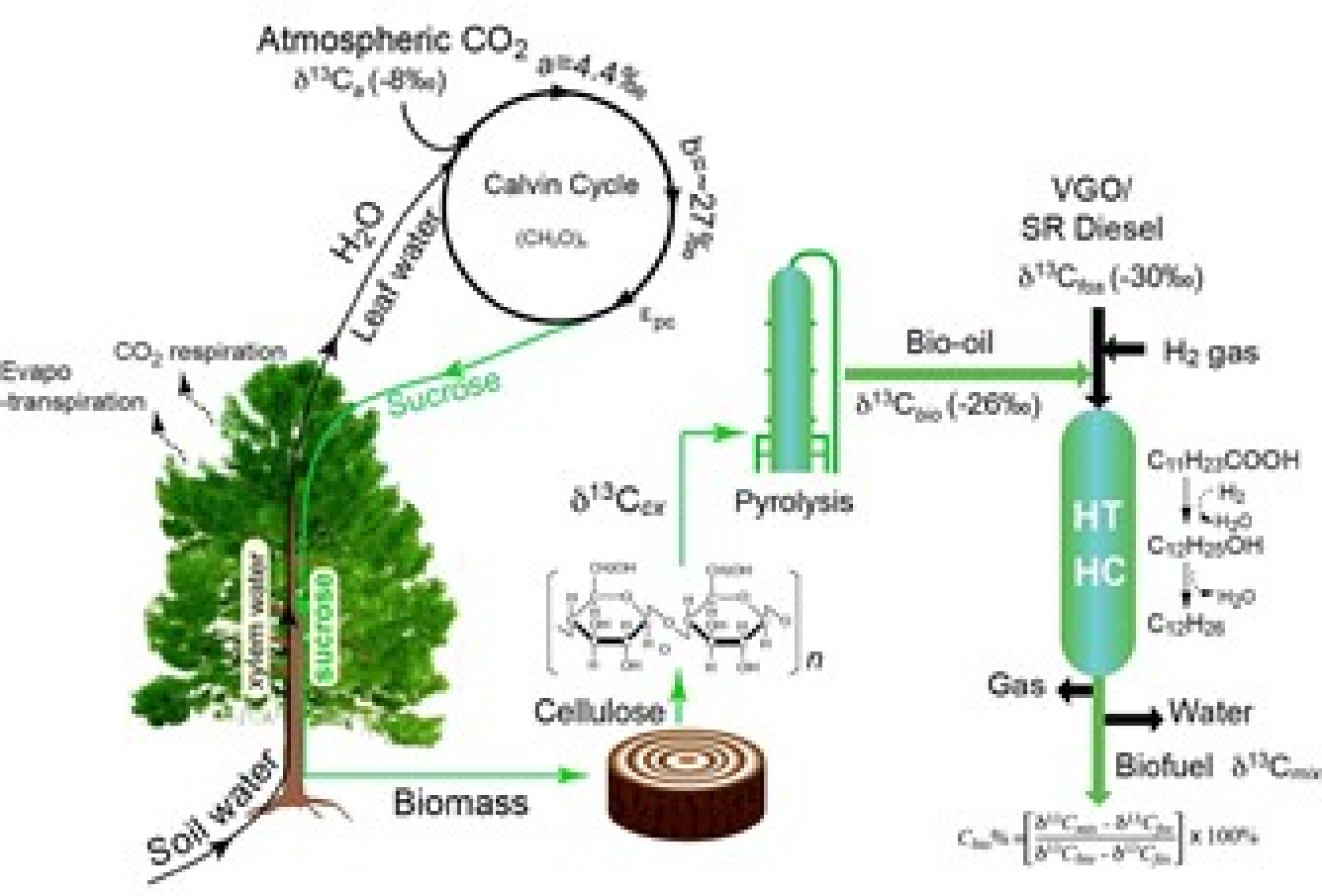
Figure 1: Using carbon isotope to track renewable carbon during the bio-oil co-processing with crude oil. Image courtesy of Los Alamos National Laboratory
ACRONYMS: VGO-vacuum gas oil, SR diesel- Straight Run Diesel, HT-Hydrotreating, HC-Hydrocracking, C11H23COOH: Lauric acid, a saturated fatty acid
A stable isotope ratio mass spectrometer (IRMS) is commonly used for determining a 13C/12C ratio in geoscience and ecology. The 13C/12C ratio in fossil oil-derived fuel and renewable material is often very small, so measurements need to be precise. However, the team discovered that standard IRMS sample preparation methods for petroleum products and bio-oils were inadequate. Oils are viscous, volatile, and contain particulate matter, so the ratio of isotopes can change after the sample is taken—a phenomenon called isotopic fractionation—impacting the accuracy of the measurements.
LANL researchers solved this problem by using special tin capsules to cold-weld seal the bio-oil and fossil fuel samples, thus eliminating the possibility of fractionation.
This was an empty link: To further improve measurement accuracyTo further improve measurement accuracy, the sealing process was performed under precise conditions that reduced contamination by atmospheric CO2. Li and his team demonstrated that their method was reliable and the correlation between the new 13C methods and current 14C methods was excellent (>0.99). This suggests that when adequate isotopic differences exist and mixing models are calibrated appropriately, the 13C method is an effective alternative with higher sample throughput and drastically lower sample cost.
Instant Feedback for Biorefiners Maximizes Renewable Carbon Content
The Los Alamos-led 13C strategy lends itself well to automation and continuous, online measurement in a co-processing refinery because of the smaller instrumentation and comparatively simple sample preparation. Such real-time monitoring would provide instant feedback to refinery operators and engineers, allowing them to precisely tune co-processing reactors to maximize renewable carbon content in fuel products and respond quickly if processing conditions drift from optimum settings.
Other Applications for the LANL 13C Method

Figure 2: Stable isotope facility at Los Alamos National Laboratory, equipped with Finnigan MAT 253 and Thermo Delta V mass spectrometers. Image courtesy of Doug Ware
LANL's 13C method also has fuel forensics applications. The method can help determine the renewable content of commercial biodiesel blends to ensure compliance with legislation requiring certain percentages of renewable content, such as for the Biodiesel Tax Credit. The 13C method could allow regulators and producers to easily determine blending levels and provide a more accurate characterization of biodiesel products.
Finally, the cold-weld seal process developed for bio-oils is appropriate for other volatile sample types and can be used to measure hydrogen, nitrogen, and oxygen isotopes using the same IRMS. This may be enormously beneficial for determining the stable isotope ratios in fluids such as water and volatile chemicals, which are of significant interest in many areas of geoscience, ecology, and isotope forensics.
Dr. Sheila Van Cuyk

Dr. Sheila Van Cuyk is the Laboratory Relationship Manager for Bioenergy Technology Office programs at Los Alamos National Laboratory (LANL). She is a scientist in the Bioscience Division, LANL’s Biofuels Program Manager in the Applied Energy Program Office, and a National Security and Defense Program Manager for Global Security.
Sheila has a background in molecular biology and environmental engineering and has over 15 years of experience developing innovative interdisciplinary solutions to complex problems. Prior to her current role, she fulfilled an Intergovernmental Personnel Act billet as a Program Manager at the Department of Homeland Security, Science and Technology Directorate working in the areas of biological threat detection and biosurveillance.
Sheila completed her M.S. and Ph.D. in environmental engineering from the Colorado School of Mines and her B.S. in biology from the College of William and Mary.


