The electron is a subatomic particle that is found in all atoms. Unlike protons, neutrons, or the nuclei of atoms, electrons are elementary particles. This means they are not made of even smaller particles. Also unlike protons and neutrons, electrons have essentially no mass. Finally, electrons differ from protons and neutrons in that they surround the nucleus instead of being part of the nucleus.
Electrons are normally bound to the nuclei of atoms. This happens because electrons have a negative charge that interacts with the positive charge of the nucleus of an atom. In a neutral atom the number of electrons is the same as the number of positive charges in the nucleus.
All the elements of the Periodic Table are neutral. However, any atom can have more or fewer electrons than positive charges. This makes the atom negatively or positively charged. These charged atoms are known as ions. More specifically, negatively charged ions are called anions and positively charged ions are called cations.
Atoms lose electrons because of how they interact with forces beyond atomic nuclei. When an electron gains extra energy, it can become excited. This can happen when an electron absorbs a photon (a packet of light) or collides with a nearby atom or particle. Electrons can gain enough energy to leave atomic nuclei behind. These free electrons mix with ions to form a plasma.
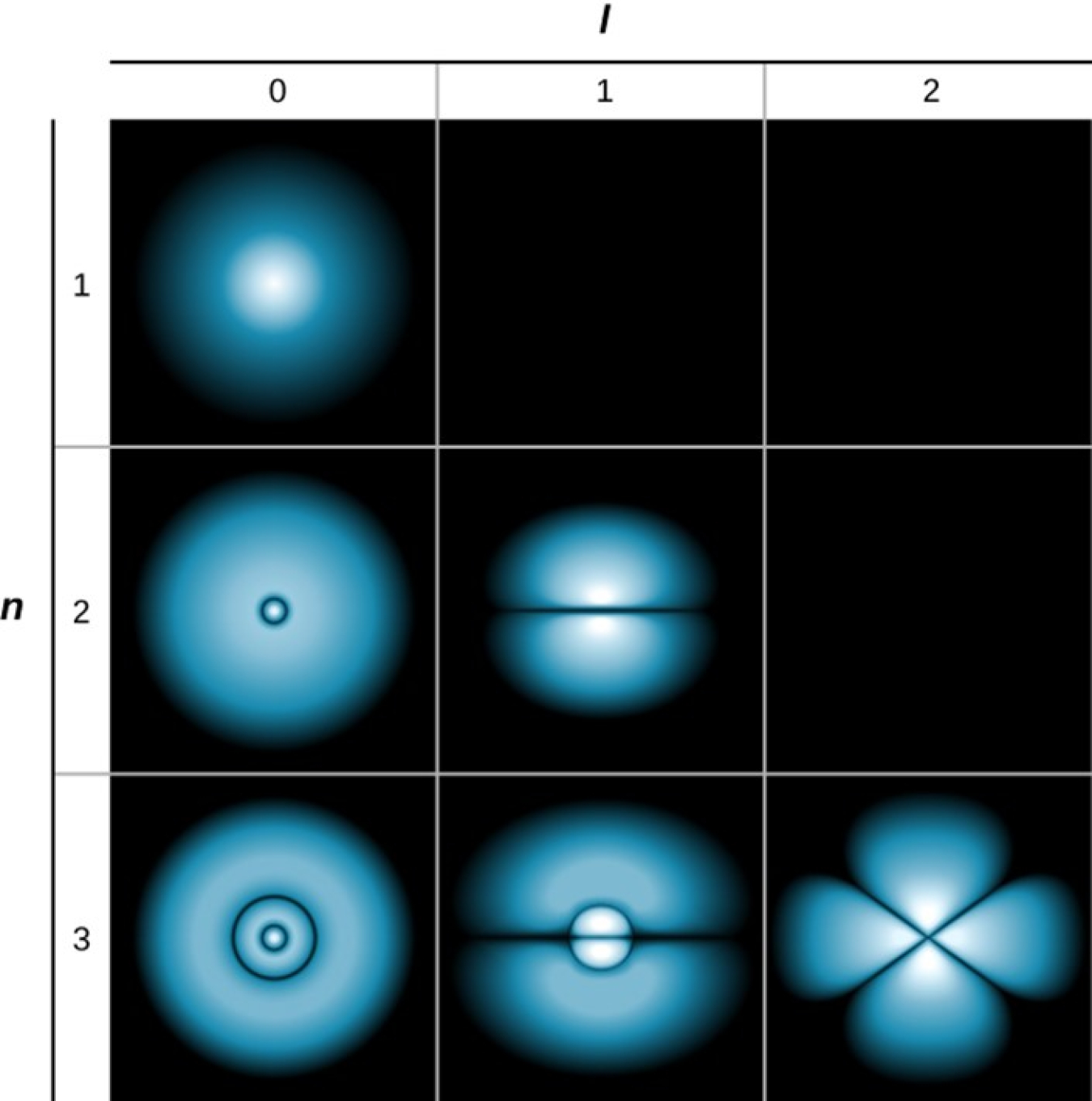
Electrons can separate from atoms because of the way they surround nuclei. In the past, scientists believed electrons orbited nuclei in the same way the moon orbits the Earth. However, scientists now believe electrons surround nuclei in a cloud divided into shells. These shells are similar to the different layers in the Earth’s atmosphere. But electrons do not exist in specific points in these shells. Because of quantum mechanics, electrons act as both points and waves. This means the areas within the shells have different probabilities of containing electrons.
Atoms have multiple shells, each with a different number of subshells. Shells can hold more and more electrons the further they are from the nucleus, and electrons have higher and higher energy levels as they move to shells away from the nucleus.
If the two outer shells are full, the element is much less reactive with other elements. This low reactivity is why the elements in the last column of the periodic table—helium, neon, etc.—are called the “noble gases” because they don’t react.
When electrons become “excited” and gain energy, then “relax” and return to their original shells, the atom emits a photon. The wavelength of the photon depends on how far the electron falls back toward the atom’s nucleus. These wavelengths are specific to each element.
DOE Office of Science: Contributions to Subatomic Particle Research
The DOE Office of Nuclear Physics in the Office of Science supports research to understand all forms of nuclear matter and the subatomic particles that make up atomic nuclei. This research includes unraveling previously unknown properties of atoms and the subatomic particles they are composed of in their natural state for important applications in medicine, commerce, and national defense. Another area of study is understanding precisely how nuclei are structured depending on the number of protons and neutrons inside them. Other research focuses on heating nuclei to the temperature of the early universe to understand how they condensed out of the quark-gluon soup that existed at the time.
Fast Facts
- Learn about electrons and other components of the elements with this interactive periodic table.
- Electrons can “tunnel” and appear on the opposite side of a barrier. It’s a phenomenon called quantum tunneling, and it’s possible because electrons are both particles and waves, and part of a wave can be on the other side of a barrier. This phenomenon is used in electron microscopes and modern integrated circuits.
Resources
- The Department of Energy Office of Science’s Nuclear Physics program
- The U.S. Particle Physics research consortium
- Science research highlight: How stiff is the proton?
- Science research highlight: The proton antimatter imbalance
Scientific terms can be confusing. DOE Explains offers straightforward explanations of key words and concepts in fundamental science. It also describes how these concepts apply to the work that the Department of Energy’s Office of Science conducts as it helps the United States excel in research across the scientific spectrum.

