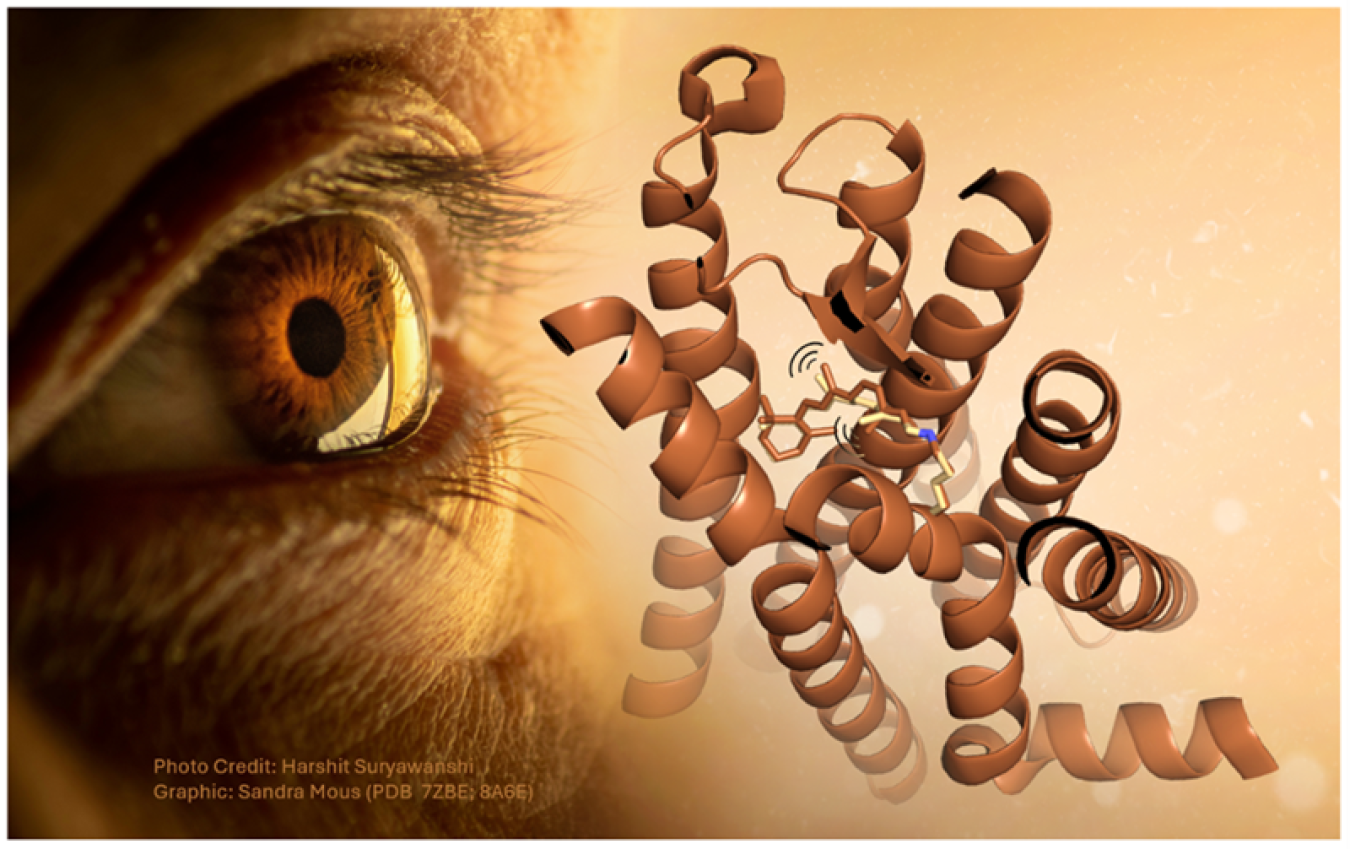
How visual rhodopsin responds to light in one of nature’s fastest reactions.
August 28, 2025The Science
Our ability to see relies on a family of proteins called rhodopsins. These receptors respond to light through an ultrafast photochemical reaction. However, scientists still don’t know the precise mechanism by which light activates the rhodopsin molecule. A team of scientists recently took the first steps towards deciphering the molecular mechanism. They made use of a powerful duo: optical laser pulses and X-ray light. First, they used a green laser pulse to activate rhodopsin in a crystalline form. Next, they used an ultrafast burst of X-ray light from an X-ray free electron laser to determine the molecule’s structure. By varying the time delay between the green laser pulse and the X-ray pulse, the team caught rhodopsin in action. This technique helped them capture the ultrafast photochemical reaction.
The Impact
By capturing the ultrafast dynamics in the rhodopsin photoreceptor, the team helped reveal the first steps leading to vision. This receptor transforms the absorption of light into a physiological signal. This signal results in electrical impulses being sent to the brain. To capture the photochemical reaction in action, the researchers used an X-ray free electron laser at the Linac Coherent Light Source, a DOE Office of Science User Facility. This is a unique experimental tool that provides ultrafast X-ray pulses. This work also demonstrates how powerful X-ray light sources, such as X-ray free electron lasers, can capture ultrafast dynamics in biology.
Summary
By studying the light-triggered dynamics of rhodopsin, the team gained insight into how light energy triggers specific motions within the receptor. While scientists had identified the initial and final structures of the receptor in a frozen state, the intermediates had so far remained elusive. These intermediate stages are critical to understanding the mechanism. The team managed to capture three of these intermediates by imaging the rhodopsin receptor at just one picosecond (one trillionth of a second), 10 and 100 picoseconds after photoactivation. The captured intermediate showed how the receptor’s chromophore absorbed the light energy and changed its shape. The chromophore is located in the middle of the receptor and makes rhodopsin light sensitive. At the same time, the scientists saw the region around the chromophore expand, making space for it to rotate and elongate. Ultimately, the changes in the shape of the chromophore and the receptor trigger a signaling cascade inside of the cell. This cascade causes the optic nerve to send an electrical impulse to the brain.
Contact
Mark Hunter
SLAC National Accelerator Laboratory
mhunter2@slac.stanford.edu
Valérie Panneels
Paul Scherrer Institute
valerie.panneels@psi.ch
Funding
This research was supported by the European Union’s Horizon 2020 program, the Swiss National Science Foundation, the Japan Society for the Promotion of Science, the Japan Agency for Medical Research and Development, the National Science Center (Poland), and the Swedish Research Council. A portion of this research used resources at the Linac Coherent Light Source, a U.S. Department of Energy Office of Science User Facility. Research also took place at the SwissFEL of the Paul Scherrer Institute, Switzerland and the Spring-8 Angstrom Compact free electron Laser (SACLA), Japan.
Publications
Gruhl, T., et al. Ultrafast structural changes direct the first molecular events of vision. Nature 615, 939-944 (2023). [DOI: https://doi.org/10.1038/s41586-023-05863-6]
Related Links
Earliest molecular events of vision revealed, Nature Magazine
How vision begins, PSI