Research on photosynthetic antenna complexes illuminates how they harvest light in plants, algae and bacteria.
November 3, 2016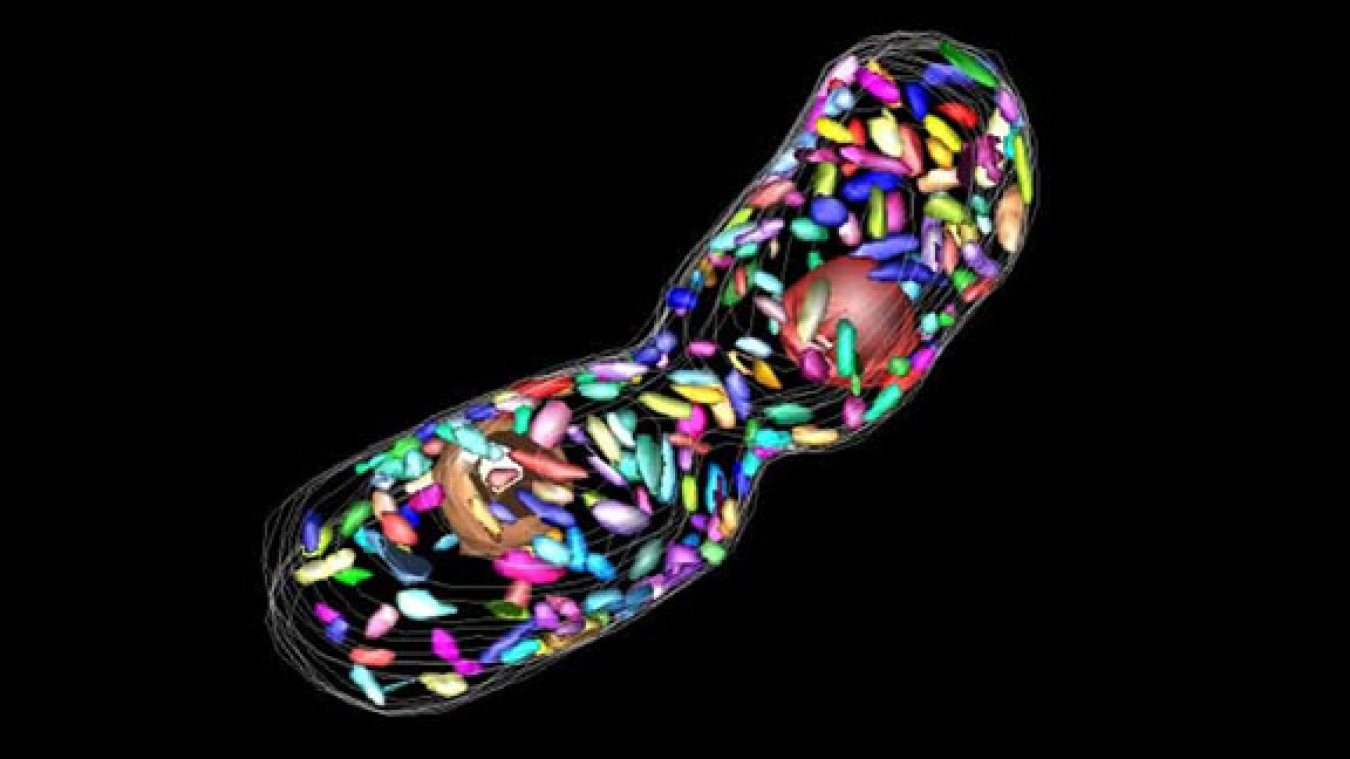
Electron microscopic tomogram of dividing cells of the green sulfur bacterium Chlorobaculum tepidum, with chlorosomes rendered in simulated color.
Walk through any woods, branches swaying overhead, and you're surrounded by billions of the world's most efficient systems for collecting energy. Inside every leaf, blade of grass, and algal cell, clusters of proteins and tiny pigments, called photosynthetic antenna complexes, capture sunshine.
Improving our understanding of antenna complexes could help scientists develop artificial systems that mimic leaves by turning sunlight into electricity or fuel. It could also lay the foundation for making the process of photosynthesis in plants, algae, and microbes more efficient. This technology could increase these organisms' yields, potentially making biofuels less expensive and easier to produce.
To expand our understanding of photosynthesis, the Department of Energy's (DOE) Office of Science (SC) supports research at the Photosynthetic Antenna Research Center (PARC), a DOE Energy Frontier Research Center. Researchers at PARC are studying how antenna complexes organize their components and protect themselves from damage.
From Photon to Photochemistry
All photosynthesis begins with a single step – a photon of light hits a pigment. The pigment (chlorophyll in plants and algae; bacteriochlorophyll in some bacteria) reacts to the photon by giving up an electron. That electron is then passed to the reaction center, where it produces products that ultimately power the plant cell.
The photosynthetic antenna complex is a collection of pigments and proteins that capture and funnel energy from light. Antenna complexes are very diverse, with their size, structures, and even types of pigments differing between organisms.
"They're remarkably different," said Robert Blankenship, a professor at Washington University and the director of PARC. "Once we understand them, we can pick and choose [characteristics], using them as inspirations for synthetic systems."
Green Sulfur Bacteria Enlighten Photosynthetic Research
The green sulfur bacteria, or Chlorobiaceae, comprise one of the most unique families of photosynthetic organisms. These peculiar bacteria live deep in the sediments of ponds, lakes, and oceans where they encounter only a few photons of light a day. In this environment, the bacteria are seldom exposed to oxygen and don't produce it.
But what green sulfur bacteria lack in exposure to light, they make up for in efficiency. They have two antenna complexes that work together: chlorosomes and the Fenna-Matthews-Olson (FMO) complex.
Chlorosomes are the largest known photosynthetic complexes in existence. While some types of antenna complexes consist of only few dozen pigments, chlorosomes are made up of as many as 250,000 pigment molecules. In addition, chlorosomes move energy much faster than their counterparts in plants and algae.
As SC-supported researchers said in a paper in the New Journal of Chemistry, "Chlorosomes are perhaps Nature's most spectacular light-harvesting antennas."
Green sulfur bacteria's unusual nature makes them ideal subjects for photosynthetic research. Their antenna complexes are both easier for researchers to build versions of than those in plants and have characteristics that could be valuable for future technologies.
Learning How Antenna Complexes Assemble
Like a metal skeleton supporting a greenhouse's windows, plants' and algae's antenna complexes have scaffolds made of proteins. These scaffolds hold pigments in place and control how pigments transfer energy between each other and the reaction center.
However, chlorosomes in green sulfur bacteria lack these scaffolds. Instead, the pigments organize themselves into stacks. Because this self-organization is simpler than the protein scaffolds, it may be easier for scientists to copy.
But because each chlorosome has a unique structure, they're difficult to study. To resolve this issue, ERFC-supported researchers developed mutated forms of green sulfur bacteria. The pigments in these altered forms were distributed more consistently than those in natural ones. The scientists found that small variations in the chlorosomes' pigment "building blocks" led to very different structures. It's like playing Tetris – each set of shapes leads to a different pattern of stacking them.
Scientists at PARC partners North Carolina State, Washington University, and the University of California, Riverside are building their own pigment molecules and have constructed 30 different chlorins (a type of pigment) from scratch. By adding and removing chemical groups, researchers could "tune" the pigments' characteristics, including the ability to stack themselves. Researchers may be able to use this technique to create synthetic light-harvesting systems.
"We're starting to understand at a molecular level how this natural system is put together," said Blankenship.
Neither Too Much nor Too Little Sun
Just like humans need to protect themselves from the sun, so do plants, algae, and bacteria. Even though photosynthetic organisms need light to grow, too much energy can damage them.
When a chlorophyll pigment normally absorbs light, it gives up an electron to the reaction center. However, if the reaction center is too busy conducting photosynthesis, it can't accept it. Instead, the pigment passes on the energy to a specialized set of proteins and pigments that can get rid of it. Researchers want to better understand this process so they can develop plants that maximize the amount of light used while minimizing damage.
Green sulfur bacteria, specifically Chlorobaculum, once again offers a unique perspective. Washington University researchers studied how Chlorobaculum's FMO antenna complex protects itself. Unlike regular plants, oxygen damages Chlorobaculum's reaction centers. To prevent harm, the bacterium slows down photosynthesis both when there's too much sun and when oxygen is present.
However, scientists knew that Chlorobaculum's FMO complex doesn't use any of the pigments that usually protect plants, algae or bacteria. Probing the complex, they expected to find some unusual amino acid, the building block of protein.
Instead, they found two completely normal cysteines, one of the 20 well-understood amino acids. Just like the pigments that usually get rid of energy, these amino acids accept electrons. However, they only do so in the presence of oxygen. In addition, while other mechanisms act as a photosynthesis on/off switch, these act more like a volume control that the cell can turn up and down.
Using this new understanding, scientists may be able to modify the bacteria that make nitrogen from the soil available for plants to use. These bacteria don't tolerate oxygen well, so improving their ability to tolerate oxygen could increase yields of food and biofuel crops.
From self-assembly to self-protection, scientists' work is illuminating how these tiny antenna complexes operate inside every green leaf you spot on a forest stroll.
The Office of Science is the single largest supporter of basic energy research in the physical sciences in the United States and is working to address some of the most pressing challenges of our time. For more information please visit the Office of Science website.
Shannon Brescher Shea

Shannon Brescher Shea (shannon.shea@science.doe.gov) is the social media manager and senior writer/editor in the Office of Science’s Office of Communications and Public Affairs. She writes and curates content for the Office of Science’s Twitter and LinkedIn accounts as well as contributes to the Department of Energy’s overall social media accounts. In addition, she writes and edits feature stories covering the Office of Science’s discovery research and manages the Science Public Outreach Community (SPOC). Previously, she was a communications specialist in the Vehicle Technologies Office in the Office of Energy Efficiency and Renewable Energy. She began at the Energy Department in 2008 as a Presidential Management Fellow. In her free time, she enjoys bicycling, gardening, writing, volunteering, and parenting two awesome kids.


