Background
Currently, the most promising alternative to thin film photovoltaic technologies that use rare elements such as CIGS and CdTe is copper zinc tin sulfoselenide (Cu2ZnSnSexS4-x, or simply CZTS). Other alternatives, such as lead sulfide (PbS) and pyrite (FeS2)-based materials, have also garnered attention. This section focuses on CZTS as a model system for Earth-abundant chalcogenide absorbers.
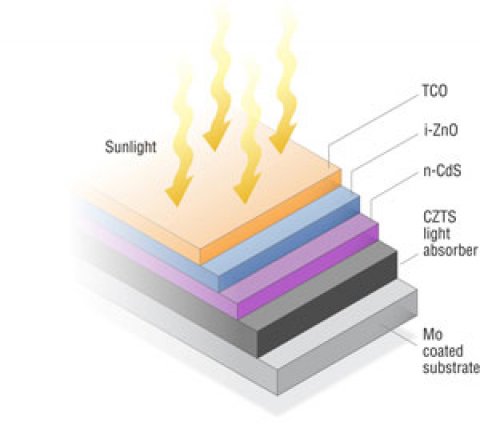
CZTS is very similar to CIGS in optoelectronic and crystallographic properties, as well as in methods of fabrication. However, CZTS has a laboratory efficiency of just above 10%, which is about half that of CIGS cells. Kesterite CZTS is very similar to the chalcopyrite crystal structure, but with the Group-III indium (In) and gallium (Ga) ions replaced in an ordered manner with an equal number of Group-II zinc ions and Group-IV tin ions. This maintains many of the optoelectronic properties of CIGS, but eliminates the need for the expensive In and Ga metals.
Research Directions
For CZTS to be commercially viable, higher efficiencies are needed. A more in-depth understanding of the defect physics of CZTS is essential, including the primary recombination pathways and to what extent the CZTS defect structure behaves like that of CIGS.
Additionally, similar to CIGS cells, CZTS cells would benefit from an optimized back contact and window layer. Researchers are investigating replacements for the CdS window layer in CZTS cells, and an optimal back-contact material.
Researchers are also developing other Earth-abundant materials, such as iron sulfide (FeS2), commonly known as pyrite, lead sulfide (PbS), and tin sulfide (SnS). These materials have exhibited some solar-relevant properties and have similarities to other solar absorber materials.
Learn more about the DOE Solar Energy Technologies Office awardees and the projects involving CZTS below.
- Purdue University (Next Generation Photovoltaics II Projects)
- Solexant (SunShot Incubator)
- University of Washington (Next Generation Photovoltaics II Projects)
- National Renewable Energy Laboratory (Next Generation Photovoltaics II Projects) – disordered CZTS
Learn more about the awardees and the projects involving non-silicon Earth-abundant solar cell technologies below.
- Massachusetts Institute of Technology (Next Generation Photovoltaics II Projects) - SnS
- University of California, Irvine (Next Generation Photovoltaics II Projects) - FeS2
- University of Wisconsin–Madison (Next Generation Photovoltaics II Projects) - FeS2
- National Renewable Energy Laboratory (Next Generation Photovoltaics II Projects)
- Developing a novel class of Earth-abundant materials for single-junction, tandem-junction, or multijunction thin-film applications
- Characterizing Earth-abundant, thermodynamically stable, and nonreactive Cu-M-N materials
- Creating a (Zn,Mg)Cu oxysulfide solar absorber material with the potential to exceed 20% energy conversion efficiency
- University of Chicago (Next Generation Photovoltaics II Projects)
Benefits
The benefits of CZTS solar cells include:
- Abundant, low-cost materials: Because of its similar thermal expansion and crystal lattice constants, CZTS can be put in a solar cell with a CdS (cadmium sulfide) window layer and molybdenum (Mo) back contact and derive about the same functionality as a CIGS cell, while using more abundant and less costly materials for the absorber layer.
- Tunable bandgap: CZTS has a tunable bandgap that can be modified from ~1 electron-volt (eV) for copper zinc tin selenide to ~1.5 eV for copper zinc tin sulfide by modulating the ratio of S to Se. Moreover, by doping the film with Ga, researchers have reached bandgaps as high as 2.25 eV.
Production
Neither non-silicon Earth-abundant nor CZTS solar cells are currently produced in large quantities. Record CZTS cells have been produced using a CZTS solution in hydrazine, which may not be favorable for manufacturing. This solution is then deposited onto a substrate and annealed. Using this process, efficiencies of greater than 10% have been achieved. Other methods that have been used for depositing CZTS include use solution processable nanocrystal inks and vacuum based deposition systems.
Similar to CIGS, including sodium appears to be important to CZTS performance, but more work is needed to fully understand this phenomenon in CZTS.
For more information on solar cells, visit the Energy Basics website.