Electrode engineering produces unprecedented selectivity, and high rates of carbon dioxide reduction to multicarbon products.
October 18, 2022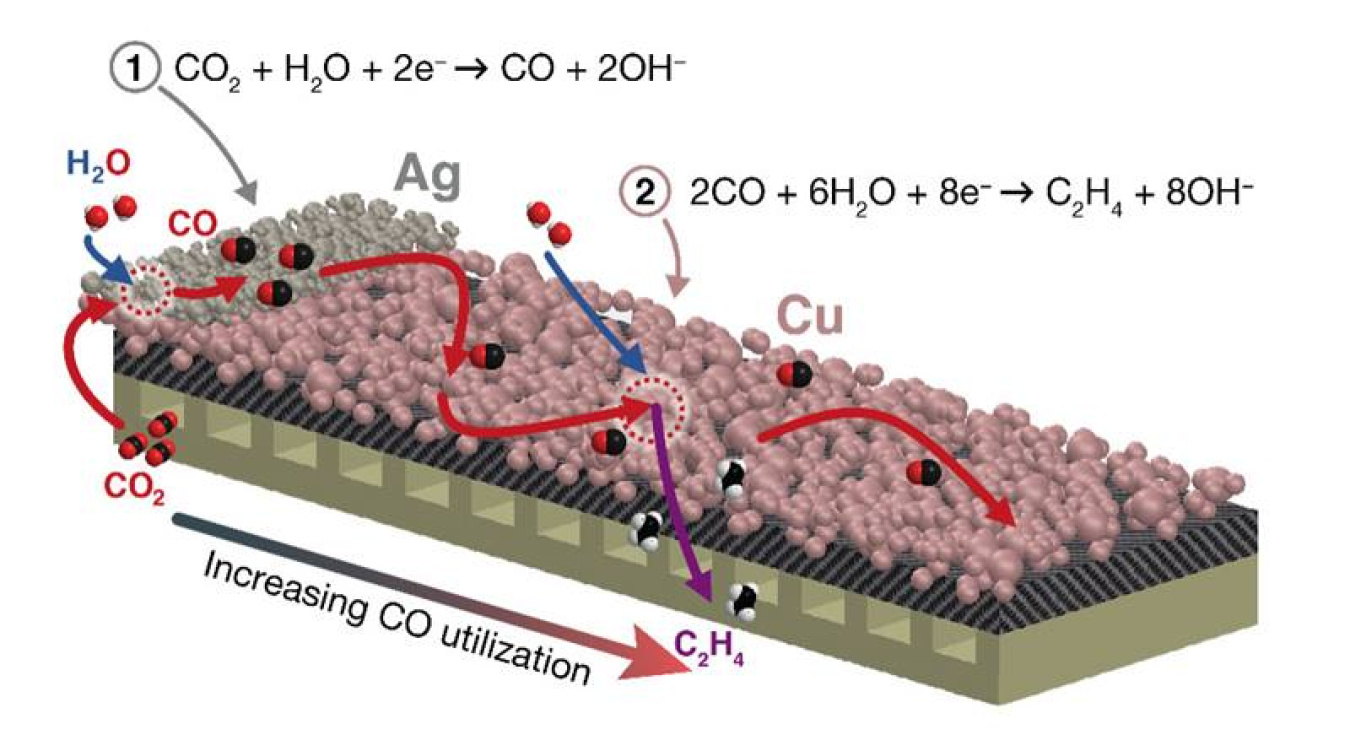
The Science
Scientists can convert carbon dioxide, a greenhouse gas, into higher value multicarbon products, seen as intermediates for potential fuels. In this research, scientists examined a new approach to this conversion process. The approach uses a material called a tandem catalyst—an electrode with two catalytic regions. The first segment uses silver or an iron-nitrogen-carbon-based catalyst to convert carbon dioxide into carbon monoxide. The second region uses a copper catalyst to convert carbon monoxide into compounds that contain multiple carbon atoms. Multicarbon products can be used as raw materials for other chemicals or potential fuels. In this new research, the scientists identified relative sizes for the two regions in the electrode and rates of carbon dioxide introduction to improve the rate of conversion of carbon dioxide to multicarbon products.
The Impact
Converting carbon dioxide from the air or from industrial emissions to higher value fuels could be a promising way to reduce greenhouse gas emissions. This approach could partially replace fossil fuels as raw materials. These investigations created an optimized dual catalyst approach that improves on current methods of converting carbon dioxide into multicarbon compounds. This approach converts more carbon dioxide into desired compounds. The research involved experiments and simulation to understand the importance of carbon monoxide and how it moves from a catalyst where it is generated to a catalyst where it is consumed to produce products. This research also demonstrates how the structure of the catalyst and the tandem catalyst concept can improve strategies to convert carbon dioxide to multicarbon products.
Summary
Electrochemical carbon dioxide reduction (CO2R) holds considerable potential for using renewably sourced electricity to convert carbon dioxide (CO2) to valuable chemicals and fuels. However, this conversion suffers from a tradeoff between rate and selectivity. Scientists believe that carbon monoxide (CO) could play a critical role as an intermediate in the CO2R process. In this study, researchers from the University of Cincinnati, Lawrence Berkeley National Laboratory, and the University of California, Berkeley, used this concept to develop a tandem electrode architecture composed of different amounts of silver (Ag) or iron-nitrogen-carbon (Fe-N-C) catalyst to produce CO and then its subsequent conversion over copper (Cu) to produce multicarbon (C2+) products. The approach incorporates several key innovations. These include the use of porous electrode architecture and gas instead of traditional liquid feeds in the cathode and a liquid feed in the anode, where oxygen is produced. Innovations also include the use of a new catalyst composed of Fe-N-C instead of Ag for even better production of CO at the front; and the management of the intermediate CO2 in a design flowfield that enhances the CO transport and residence time. Experiments coupled with simulation demonstrate how the ratio of Cu to Ag areas, catalyst loading of each catalyst, and gas flow rate are used to manage the transport of CO in order to obtain an optimal formation percent and rate of C2+ products.
Contact
Adam Weber
Senior Scientist, Lawrence Berkeley National Laboratory
azweber@lbl.gov
Jingjie Wu
Professor of Chemical and Environmental Engineering, University of Cincinnati
jingjie.wu@uc.edu
Funding
This material is based on work supported by the Department of Energy Office of Fossil Energy and Carbon Management and performed at the Joint Center for Artificial Photosynthesis, a DOE Energy Innovation Hub, supported through the Office of Science. This research also drew on support for students from the National Science Foundation and the National Defense Science and Engineering Graduate Fellowship Program sponsored by the Army Research Office.
Publications
Zhang, T., et al., Highly Selective and Productive Reduction of Carbon Dioxide to Multicarbon Products via in-situ CO Management using Segmented Tandem Electrodes. Nature Catalysis 5, 202–211(2022). [DOI: 10.1038/s41929-022-00751-0]

