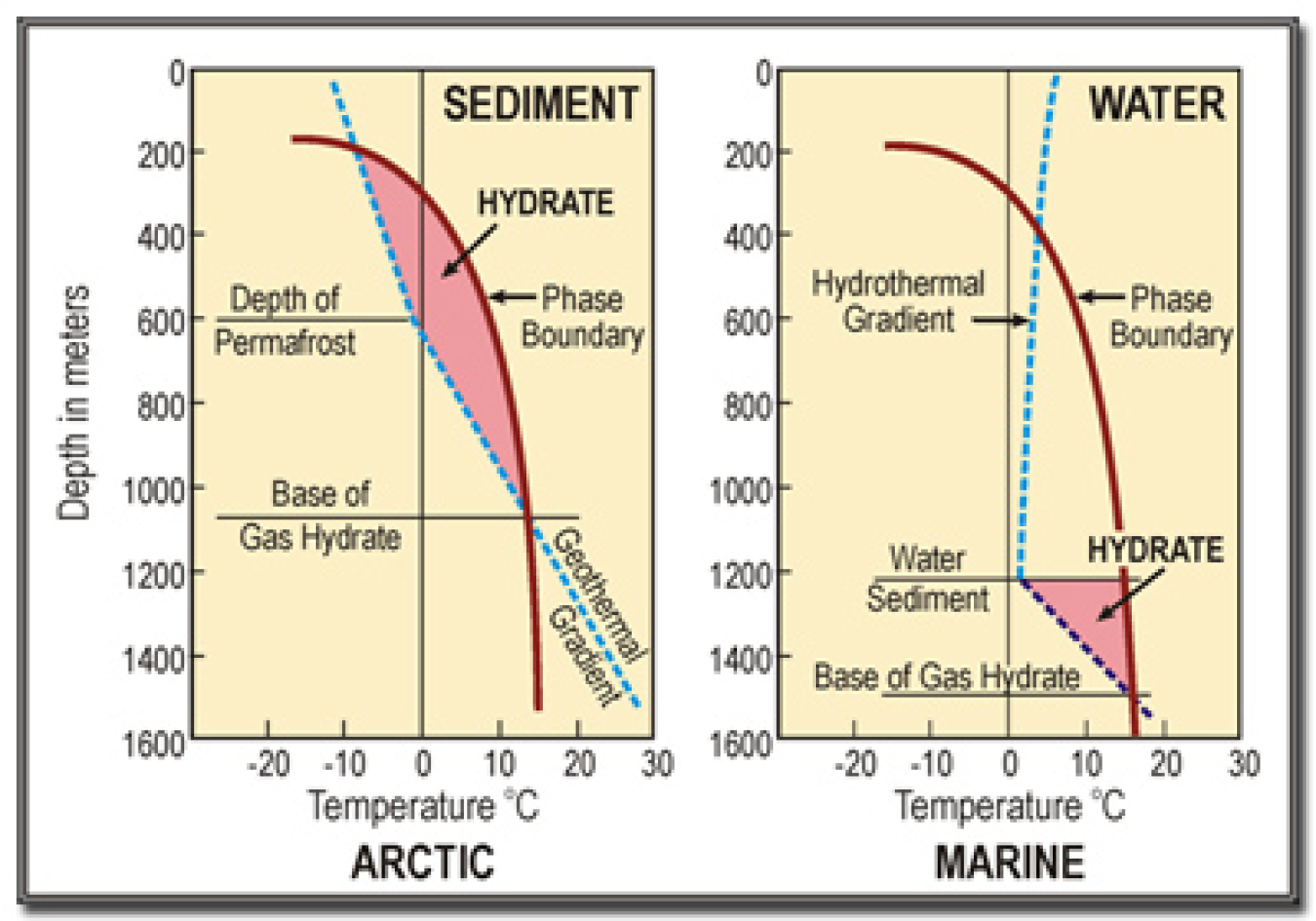
The red curves are temperature profiles for various water depths; the blue line shows methane hydrate stability relative to temperature and pressure. The area enclosed by the two curves represents the area of methane hydrate stability.
Methane, the predominant component of natural gas, forms hydrate in the presence of water, low temperatures and high pressures. Alternatively, when the temperature is increased or the pressure decreased so that hydrates are outside their stability field, they dissociate into methane and water. Laboratory studies, modeling and short-term field programs have shown that methane production is technically feasible using depressurization. However, there has not been a long-term demonstration of methane production from hydrate.
DOE's methane hydrate program is focused on developing the technology and knowledge that will allow industry to commercialize methane hydrate production. Our field programs are focused on the Alaska North Slope and offshore Gulf of Mexico as both contain prolific known petroleum systems conducive to the formation of gas hydrates, as well as a wealth of information and infrastructure developed during decades of industry hydrocarbon exploration. The well-delineated gas hydrate occurrences on the Alaska North Slope provide relatively low-cost opportunities to conduct extended duration field production trials. The Gulf of Mexico provides a laboratory for testing gas hydrate exploration technology and providing an initial confirmation of the scale of marine gas hydrate occurrence in the United States. Because methane hydrate resources are predominantly located offshore, the Gulf of Mexico is the ultimate target for technologies first tested in the arctic.

