Bioprose Blog: A New Source of Renewable Plastics: Converting Ethanol to Butadiene in a Single Step
January 23, 2018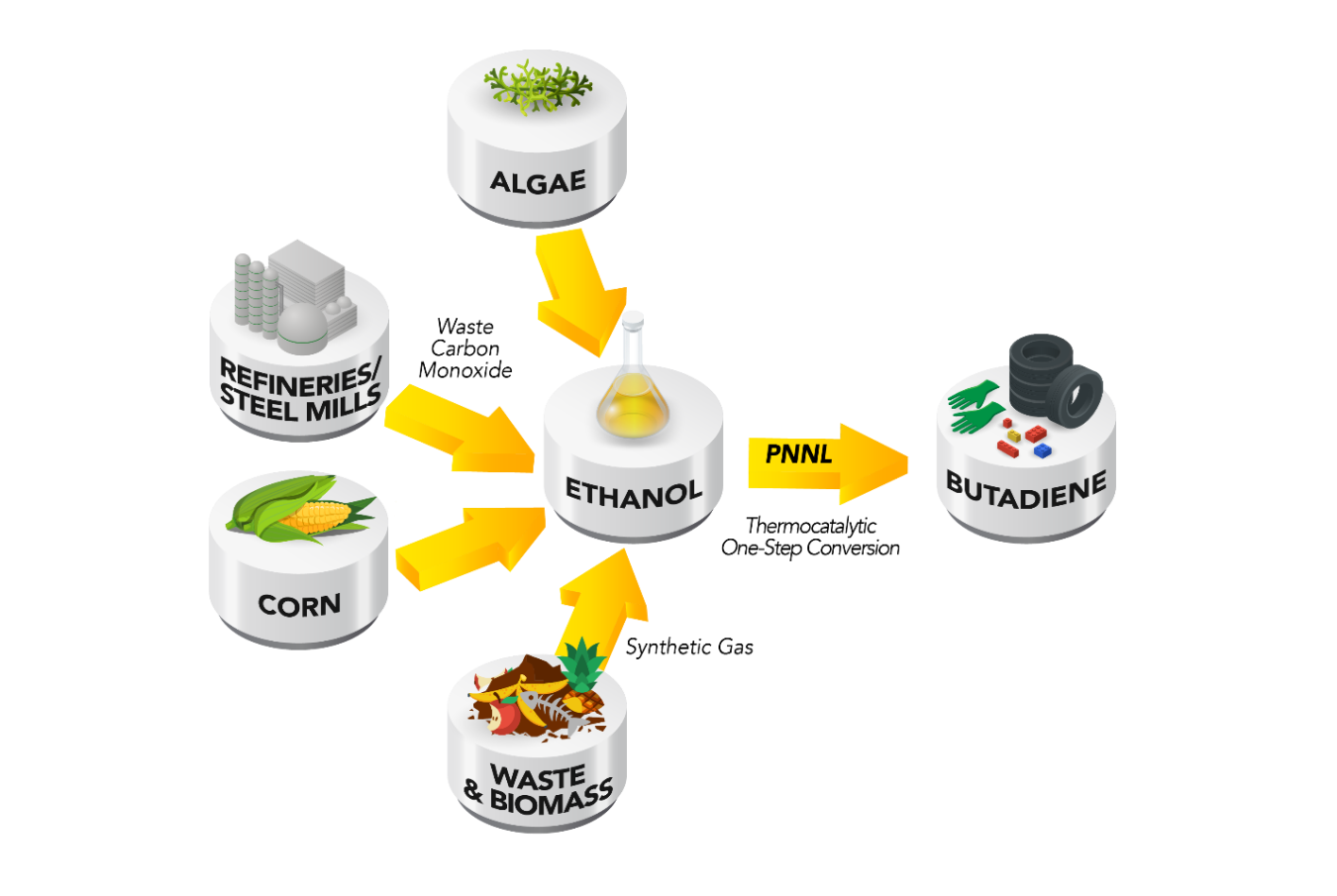
Researchers at Pacific Northwest National Laboratory develop a new catalyst capable of converting ethanol to butadiene, opening the door to a renewable source of polymers and plastics.
Author: Corinne Drennan, Laboratory Relationship Manager, Pacific Northwest National Laboratory
Read Corinne's bio ►
Meet the other bloggers ►
Return to Bioprose blog ►
Bioprose Blog
If America wore a pair of shoes, the soles would be made of butadiene. This molecule is the building block for just about every major synthetic plastic or rubber. Tires: styrene-butadiene. Fuel hoses: nitrile-butadiene. Children’s toys: acrylonitrile butadiene styrene. It’s important stuff, and researchers at Pacific Northwest National Laboratory (PNNL) just developed a catalyst capable of converting ethanol directly into butadiene.
Traditionally, butadiene is produced from petroleum via a refining process known as steam cracking. In the past, refineries used heavy crude oil, which produced high yields of butadiene. However, with the rise in shale-oil or light-weight crude oil, butadiene production volumes are in danger of dropping while demand continues to rise.
That’s where PNNL steps in. As part of a project sponsored by the U.S. Department of Energy’s Bioenergy Technologies Office, researchers at PNNL are working on ways to generate butadiene from renewable sources by developing a new catalyst that can convert ethanol into butadiene.
While ethanol-to-butadiene catalysts aren’t particularly new, previous catalyst systems weren’t commercially viable. First, the catalysts were not active, so the ethanol had to be passed through the catalyst multiple times or operated at very low throughputs to reach a 70% yield—the amount of ethanol converted into butadiene. Second, previous catalysts required pure ethanol—ethanol without any water in it. Separating water from ethanol adds an additional step and cost to the overall process.
However, PNNL scientist Vanessa Dagle and her team recently developed a single-step catalyst that is capable of converting about 70% of ethanol to butadiene in a single pass and at industrially relevant process conditions. The catalyst also does not require pre-preparation of the ethanol; aqueous ethanol (ethanol with water in it) can be used.
The catalyst uses silica as a support material, with silver nitrate powder and zirconyl nitrate as the catalytic materials. It works by ripping hydrogen off the ethanol molecules, creating acetaldehyde. Carbon-to-carbon bonds form, producing crotonaldehyde, which is then converted to crotyl alcohol. The alcohol undergoes dehydration, and voila, you have butadiene.
As far as Dagle knows, the PNNL-developed catalyst is the most active ethanol-to-butadiene catalyst reported. To get such an efficient catalyst, the team had to balance the components of the catalyst just right—especially its acidity. Too much acidity, and ethylene—not butadiene—would be produced. Too little acidity, and the team would be left with unreacted acetaldehyde.
PNNL’s catalyst introduces the possibility of ethanol as a source of butadiene in addition to petroleum. The ethanol can be derived from renewable sources, such as corn, switchgrass, algae, and even waste gas from steel mills. If successful, the ethanol-to-butadiene catalyst could be a boon to the $23 billion butadiene market and could open the door to a new renewable source of plastics.
Dr. Asanga Padmaperuma

Dr. Asanga Padmaperuma is a Senior Research Chemist in the Energy Processes and Materials Division at Pacific Northwest National Laboratory. He serves as the Laboratory Relationship Manager for the Department of Energy's Bioenergy Technologies Office, which has oversight of research in renewable use of marine, terrestrial, and waste biomass for fuels and chemicals via thermal, biological, and electrocatalytic conversion.
Dr. Padmaperuma also serves as the Technical Leader for the Chemical Conversion team in PNNL's Chemical and Biological Processing Group. Prior to his current assignment, his research focus was on thermochemical, low-temperature catalytic, and electrochemical production of value-added chemicals and fuels from bio-based and waste sources. He also conducted research in design and development of functional molecules for organic electronics and liquid scintillators for radiation detection.
Dr. Padmaperuma completed his B.Sc. in Chemistry at the University of Colombo (Sri Lanka) and obtained his Ph.D. in Organic Polymer Chemistry at the University of Southern California's Loker Hydrocarbon Research Institute. He is a named inventor on several granted patents as well as an author of more than 50 journal articles, book chapters, and public technical reports.
Meet our other bloggers ►
Return to Bioprose blog ►



