Bioprose Blog: It’s Elemental—Designing the Perfect Biofuel Catalyst
February 8, 2018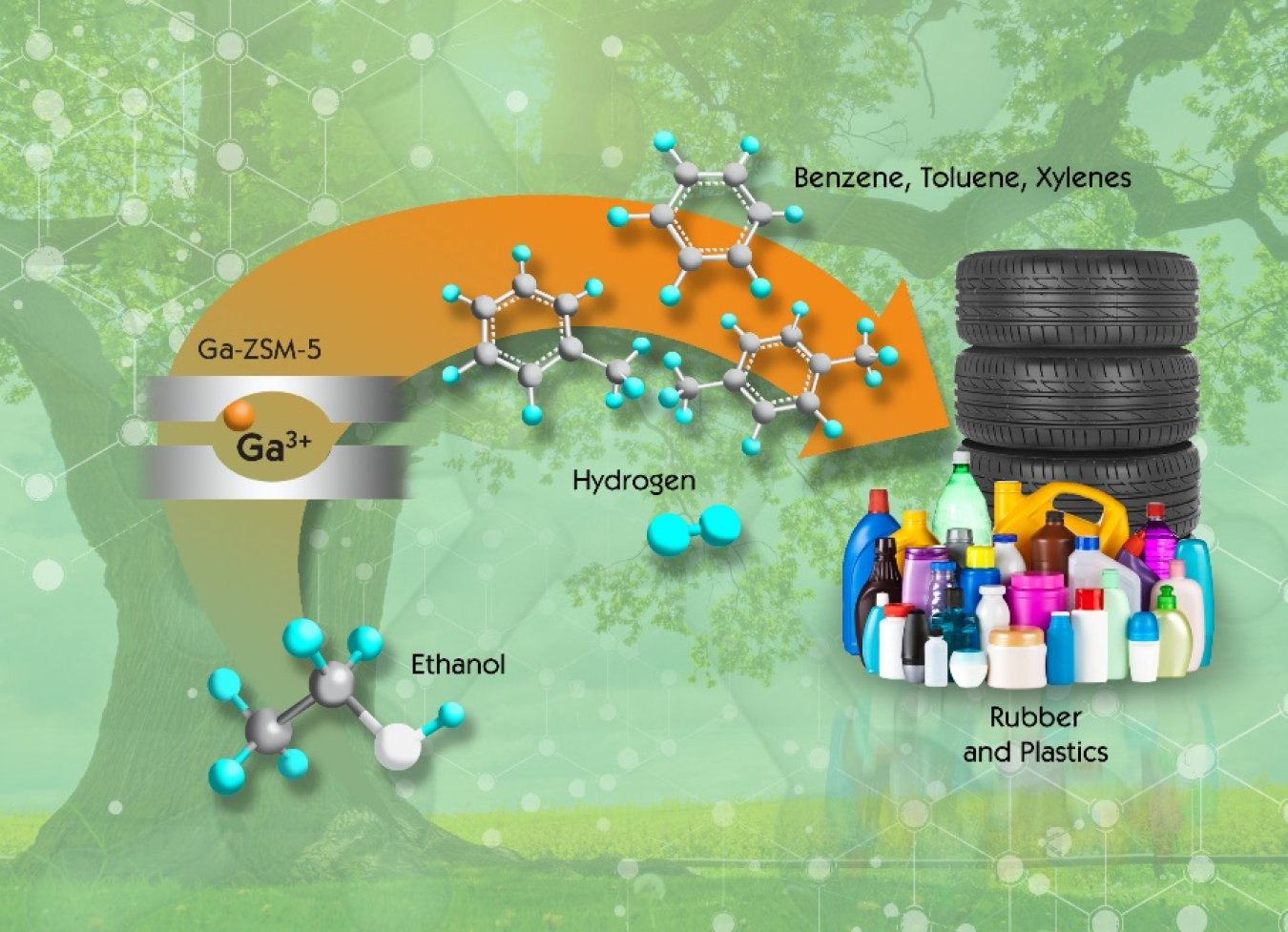
Scientists at Oak Ridge National Laboratory created a new catalyst production process that doubles the output of renewable BTX, a group of high-value chemicals that can be used to produce a range of everyday items, from soda bottles to tires.

Author: Tim Theiss, Laboratory Relationship Manager, Oak Ridge National Laboratory
Read Tims bio ►
Meet the other bloggers ►
Return to Bioprose blog ►
Bioprose Blog
The production of renewable fuels and products is getting a boost from Group 13 on the periodic table.
Several years ago, researchers at Oak Ridge National Laboratory (ORNL) developed a novel process for converting renewable ethanol into a hydrocarbon blendstock. This renewable blendstock can be mixed with gasoline, diesel, or jet fuel, as well as converted into benzene, toluene, and xylenes (BTX)—commodity chemicals commonly used to make plastics and rubber. Now, ORNL scientists have discovered the key to doubling the production of renewable BTX by modifying the highly versatile zeolite catalyst.
Perfecting the "Boiling Stone"
Zeolites officially hit the scene in 1756, when Swedish geologist Axel Cronstedt coined the name from the Greek word for "boiling stone." Many occur naturally as minerals, while others are synthetic and are made for specific commercial applications. Zeolites were in use long before the term was coined; in fact, natural zeolites were first used by the Romans to filter their drinking water. Since then, they have found application in a wide variety of industries, ranging from petroleum refining to detergent manufacturing.
The secret behind zeolites’ commercial success lies within their structure. Zeolites are crystalline solids made of silicon, aluminum, and oxygen, but most importantly, they form a framework with small cavities and channels inside that are ideal for chemical reactions—like converting biomass into fuels and products.
Building upon past successes, ORNL set out to discover how to optimize this inner framework. The key to optimization, ORNL researchers soon discovered, comes from a unique metallic element—gallium. When scientists added gallium cations to zeolites, they saw a two-fold increase in BTX yield
And That’s Not All...
The research team also developed a new method to make the zeolite catalysts. Most BTX produced from ethanol today uses a catalyst prepared with more expensive materials and involves a multi-step process that requires water. The ORNL process simplifies the catalyst synthesis, eliminating the need for water and reducing catalyst production costs.
Coming to a Store Near You
BTX is used in a wide range of industrial applications and has a higher profit margin than biofuels.
Biorefineries using this process can create an additional revenue stream by recovering BTX during conversion and selling it to plastic and rubber manufacturers for use in everyday products, like soda bottles and tires. In fact, Vertimass LLC, a California-based startup company, has licensed ORNL’s technology and is currently working on scale-up.
Want to learn more? Read about the science behind this conversion technology and check out ORNL’s recently published paper to learn how gallium cations are optimizing biofuel and bioproduct production. This work was funded through the Bioenergy Technologies Office’s Conversion Research and Development Program, which focuses on broadly addressing technical challenges to achieving affordable biofuels and bioproducts

