X-ray imaging shows that selectively etching surface nickel from a nickel-platinum alloy leaves a chemically active platinum coating.
July 26, 2023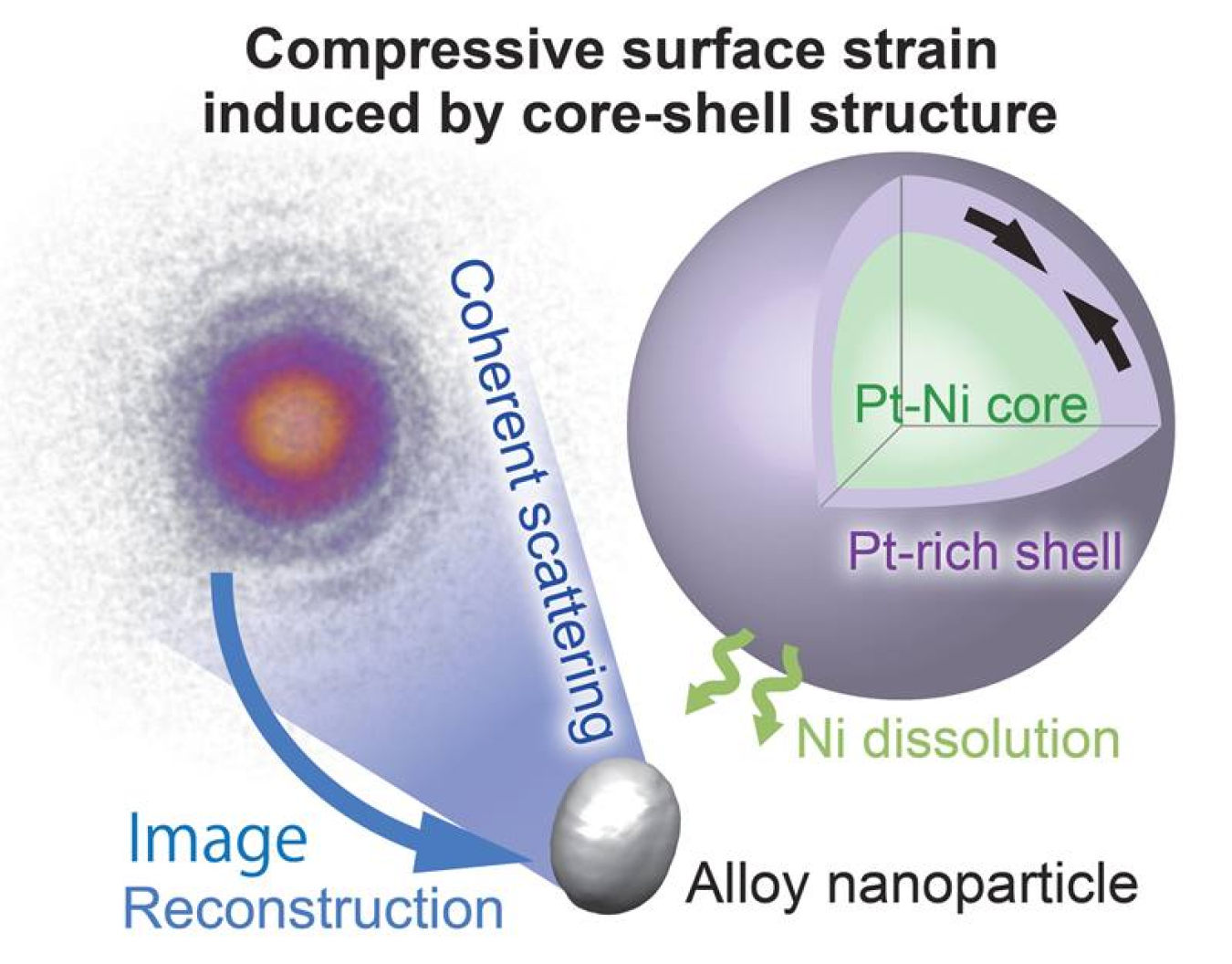
The Science
Platinum (Pt) can act as a catalyst to make reactive oxygen ions for many applications. In this research, scientists used a method called electrochemical cycling to modify the surface of nickel (Ni)/Pt nanoparticles. The scientists then examined the particles using a specialized X-ray scattering imaging technique that is uniquely suited to probe three-dimensional particles in liquids. This revealed that the modified alloy had a Pt rich layer. This layer’s structure left Pt at the surface of the nanoparticles, more concentrated than would be normal in a bulk Ni-Pt alloy. The technique reveals the composition, shape, and strain of nanometer scale particles that are used in electrodes and membranes.
The Impact
The oxygen reduction process is essential in many applications. This includes the electrodes of fuel cells, which electrochemically consume fuels directly into electricity. This also includes metal-air batteries that produce electricity by oxidizing metals. Pt can drive these reduction reactions. Replacing Pt components with alloys and improving the activity through surface treatments will make such processes less expensive and more efficient. The X-ray technique reveals how the material changes under operational conditions. Researchers can use this technique in reactive environments to evaluate the surface state of essential materials. This will help them study and improve materials for energy and chemical conversion devices.
Summary
Researchers at Argonne National Laboratory, Safarik University in Slovakia, and Tohoku University in Japan used Bragg coherent diffraction imaging (BCDI) to monitor atomic level strain on the surfaces of Pt-Ni nanoparticles while they were being treated electrochemically. This method allows researchers to determine shape, composition and atomic spacings in the actual environments where a material is processed or deployed. They monitored elastic strain during successive voltametric cycles in a liquid electrolyte as a function of Ni dissolution, as deduced from three-dimensional images from BCDI and from measurements of the average lattice constants. The results show that higher levels of initial Ni composition resulted in more dissolution and higher levels of compressive strain at the surface. The processing resulted in a core-shell structure with a Pt-rich shell surrounding a Ni-rich core. These results help explain why oxygen molecules can be more readily turned into reactive ions on Pt-Ni nanoparticles compared to pure Pt nanoparticles. The strain correlated with the dealloying can change the shape and electronic structure of absorption sites important to oxygen charge transfer.
Contact
Hoydoo You
Argonne National Laboratory
You@anl.gov
Funding
The X-ray measurements and electrochemistry were performed at Argonne National Laboratory and were supported by the Department of Energy (DOE) Office of Science, Office of Basic Energy Science, Materials Sciences and Engineering Division. The use of the Advanced Photon Source was made available by the DOE Office of Science Scientific User Facilities Division. Data analyses at Tohoku University were supported by the Center for Computational Materials Science, Institute for Materials Research. Sample preparation at Safarik University was supported by VEGA and grants from the Slovak Research and Development Agency.
Publications
Kawaguchi, T., et al.,Electrochemically Induced Strain Evolution in Pt–Ni Alloy Nanoparticles Observed by Bragg Coherent Diffraction Imaging. Nano Letters, 21, 5945–5951 (2021). [DOI:10.1021/acs.nanolett.1c00778]

