-- This project is inactive --
The University of California, Los Angeles (UCLA), through the Concentrating Solar Power: Efficiently Leveraging Equilibrium Mechanisms for Engineering New Thermochemical Storage (CSP: ELEMENTS) funding program, is addressing the use of the reversible ammonia dissociation reaction as a thermochemical energy storage mechanism for solar thermal power. One of the key aims of the research is to design and build a synthesis reactor that is optimized for the direct production of supercritical steam at 650°C.
Approach
Ammonia synthesis reactors have not previously been designed to produce supercritical steam at 650°C. It is necessary to carefully design the geometry, flow conditions, and use of catalyst in the high temperature reactor entry region, where the nearly pure N2 + 3H2 enters and must follow a carefully controlled temperature-enthalpy path as it reacts and transfers heat to the steam. The design process requires iteration between modeling and small-scale experiments to develop confidence in the model results and establish a design that will achieve the desired pilot-scale reactor behavior. The pilot-scale reactor is intended to generate 25 kWt to heat steam at 26 MPa from 350°C to 650°C, corresponding to a steam mass flow rate of 0.0125 kg/s.
Innovation
Ammonia synthesis reactors have not previously been designed to produce supercritical steam at 650°C. Furthermore, since the ammonia thermochemical energy storage system stores solar energy in gaseous hydrogen and nitrogen, which by their nature are harder to store cost effectively than liquids, this project will evaluate technology adapted from the natural gas and oil industries that could provide proven, cost effective solutions for materials storage.
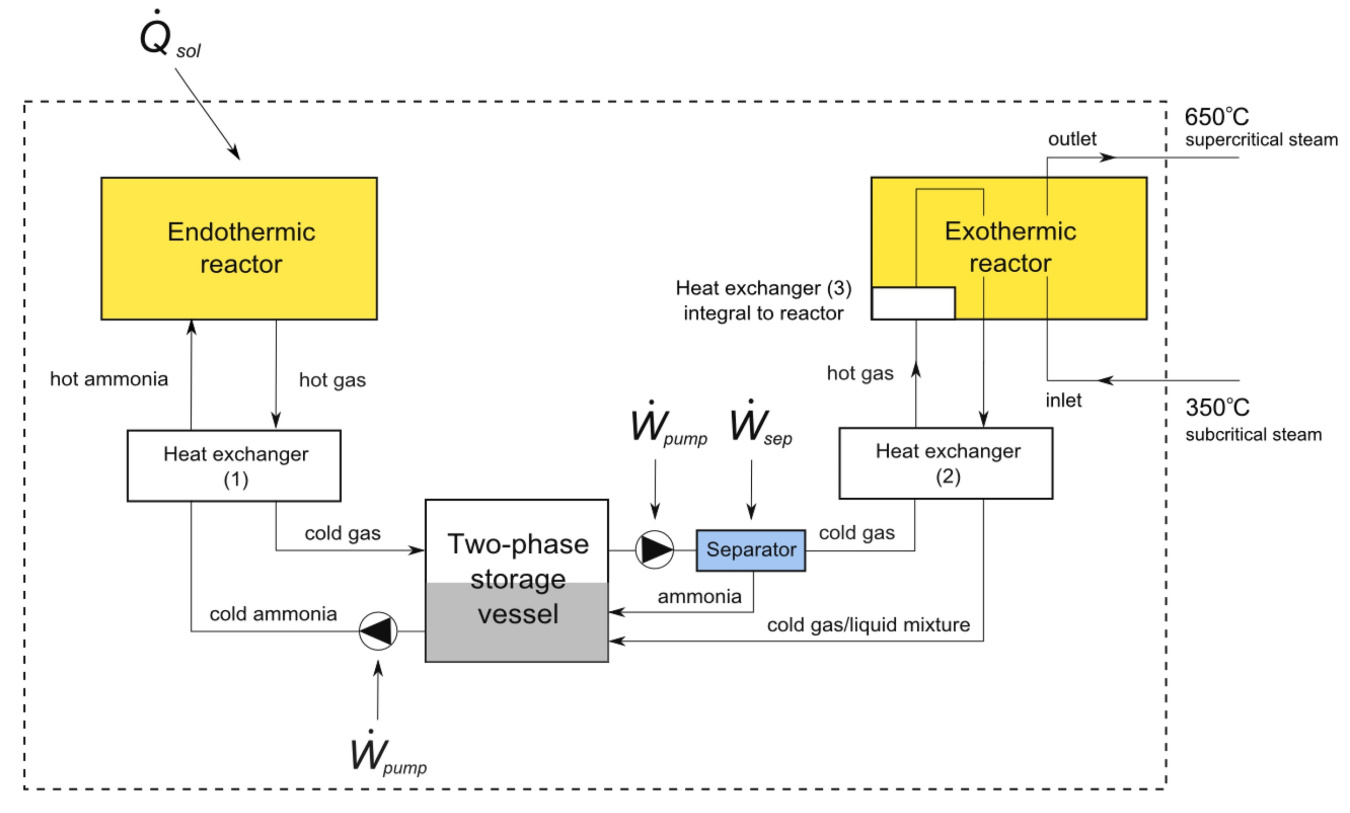
The anhydrous ammonia thermochemical energy storage cycle proposed by UCLA. The system is charged when ammonia, NH3, is heated in the Endothermic reactor by the sun (Qsol) transforming it into N2 and H2. These gasses are then cooled and stored in the two-phase storage vessel. When the sun does not shine, N2 and H2 are withdrawn from storage and passed through the exothermic reactor, which UCLA will be designing and optimizing for the direct production of super critical steam at 650°C.
Learn about other concentrating solar power research.

