-- This project is inactive --
The University of Florida (UF), through the Concentrating Solar Power: Efficiently Leveraging Equilibrium Mechanisms for Engineering New Thermochemical Storage (CSP: ELEMENTS) funding program, is working on making concentrated solar power economically competitive with traditional forms of energy generation by driving the cost towards $0.06/kWh through the use of thermochemical energy storage (TCES). The project uses inexpensive, safe, and non-corrosive strontium-based carbonates and high temperatures from concentrated sunlight to break chemical bonds and store energy during the day time. During off-sun periods, the reaction is reversed to remake the chemical bonds previously broken, releasing the stored heat, and doing so at temperatures previously unachievable in other carbonate-based TCES processes.
Approach
The viability of the strontium oxide/ strontium carbonate chemistry as a candidate for TCES will be thoroughly demonstrated. In a continuation of preliminary experiments already conducted at UF, a rapid screening of materials prepared via the technique known as Sacrificial Pore Formation using Graphite (SPF-G) will be carried out to determine if this strategy shows the potential to increase capacity and preserve stability over 2000 cycles using TGA. Fundamental measurements of the reaction kinetics for the unexplored carbonation step will yield input data for both pore level simulation and continuum modeling efforts. Building off of these small scale experiments and pore level modeling, UF will demonstrate and simulate the TCES process at the 50 g scale. Using this information, a technoeconomic analysis will estimate the probability of reaching a total capital investment of less than $10/kWhth. These economic projections will be shared with interested parties to aid in developing strategic partnership agreements.
Innovation
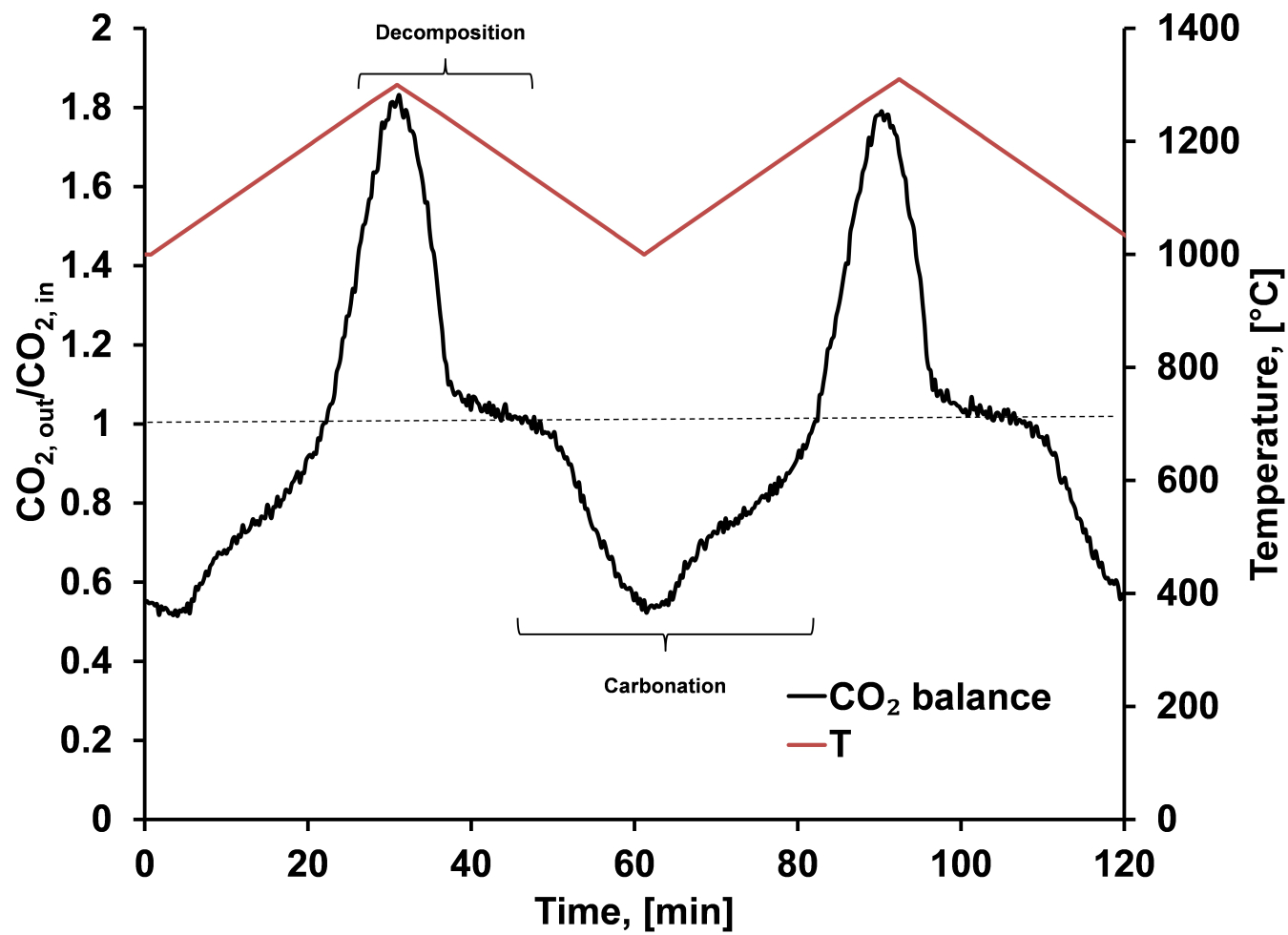
This use of strontium in conjunction with carbonate allows the system to be charged via the reversible decarbonation reaction at solar energy input temperatures of 1300°C. This, in turn, allows for discharge of the stored solar energy at very high temperatures of over 1000°C. Furthermore, the use of sacrificial pore formation may help to enhance the capacity and increase the stability of the system. Finally, the high temperatures associated with this reaction can, in theory, allow for higher efficiency power generation.
Experimental data describing the cyclic TCES process for strontium carbonate. Heating strontium carbonate up from 1000°C to 1300°C causes the compound to undergo a decomposition reaction which results in the capture of thermal energy, shown here as the peak in the black curve occurring between Time = 20 and Time = 40 minutes. Cooling the system back down to 1000°C leads to reconstitution of the starting material in a carbonation reaction thereby releasing the stored thermal energy, a process shown as the valley in the black curve between Time = 50 and Time = 80 minutes. The process is repeatable.
Learn about other concentrating solar power research.

