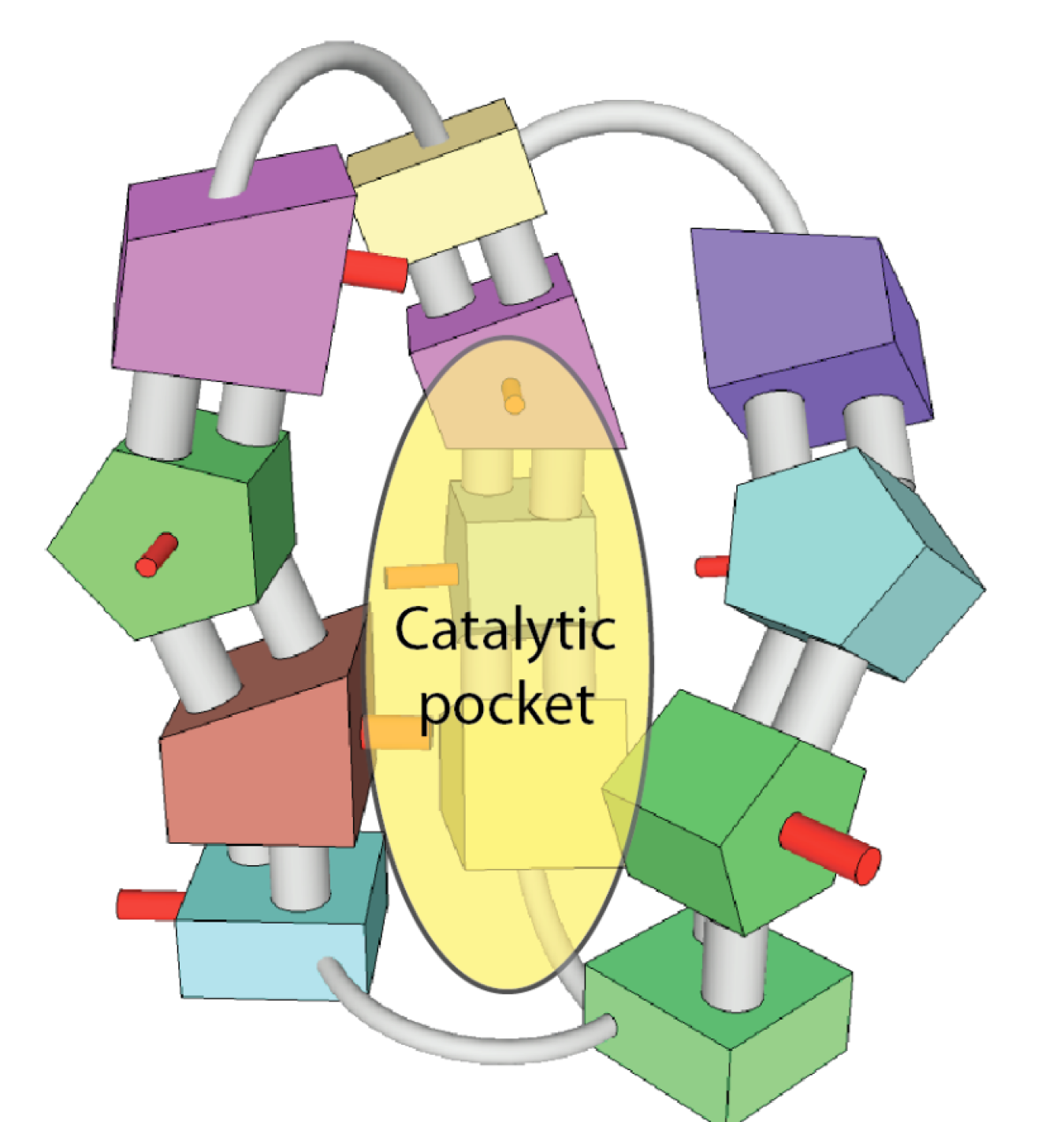
Atomically precise catalysts can have a rigid shape which mimics the best catalysts found in nature (enzymes) and create a selective “pocket” into which the chemical reactants fit.
In June 2016, five projects were selected for Phase I awards in the new Advanced Manufacturing Office (AMO) focus area of Atomically Precise Catalysts through the combined Small Business Innovation Research (SBIR) and Small Business Technology Transfer (STTR) program. Each of these small businesses will receive $150,000 for research and development of revolutionary new catalysts which will play a critical role in making manufacturing processes more energy-efficient. Catalysts accelerate the rate of reaction and can also allow for lower reaction temperatures for significant energy savings. Traditionally, catalysts are solid state materials that bring reactant molecules together, or temporarily hold them on a surface, but still rely on the random nature of molecules bumping into each other with the correct orientation and sticking to favorable sites. Atomically precise catalysts work more like enzymes in biological systems, which are highly efficient, and can be engineered with reproducible structures to “hold” the molecules together in just the right way. Reaction rates that are a billion times faster than with traditional catalysts are possible with this bio-mimicry approach.
As one project example, Proton Energy Systems, based in Wallingford Connecticut, is partnering with the University of Arkansas and Case Western Reserve University to develop an atomically precise catalyst to be used in the manufacture of ammonia. Ammonia production consumes 1-2% of world-wide energy, and the production of ammonia accounts for approximately 1% of the world’s greenhouse gas emissions. Ammonia is an important chemical component in fertilizers, and therefore is directly involved in farming and food production around the world. By reducing the amount of energy it takes to make ammonia, we can reduce the amount of energy it takes to feed the world. This is especially important in poor countries where farming is a large part of the local economies. Researchers have spent decades trying to make ammonia production more efficient, and now the team at Proton Energy Systems is working to develop atomically precise catalysts to mimic the way nature makes ammonia and apply them to industrial-scale reactions. This research has the potential to significantly reduce the amount of energy required to manufacture ammonia worldwide and reduce greenhouse gas emissions associated with ammonia production.
Another example is Mainstream Engineering, based in Rockledge, Florida, who is partnering with Temple University to design and synthesize catalysts which are put together piece-by-piece like Legos®. A variety of different molecular building blocks can result in millions of different shapes, depending on how they are put together. These Lego-like catalysts have a rigid structure designed to temporarily hold specific chemical reactants and more closely mimic the selectivity of catalysts occurring naturally in nature. If catalysts are more selective, it will reduce the need for recovering unreacted chemicals and removing byproducts. This technology also has the potential to reduce the reaction temperature required to produce the final chemical product. All of these improvements can greatly reduce the energy required for chemical production. The platform technology being developed in this project will be applicable to many manufacturing sectors such as pharmaceuticals and the flavor, fragrance, and agrochemical industries and can help reduce energy consumption and costs throughout these sectors.
These two projects along with the others in the focus area of Atomically Precise Catalysts have the potential to greatly increase the energy-efficiency of chemical manufacturing in the U.S.
To learn more about AMO’s projects through the SBIR/STTR program, click here.

